>Corresponding Author : Salah Hasab Mahmoud
>Article Type : Review Article
>Volume : 6 | Issue : 1
>Received Date : 09 March, 2026
>Accepted Date : 18 March, 2026
>Published Date : 30 March, 2026
>DOI : https://doi.org/10.54289/JDOE2600103
>Citation : Ashraf S, Elawsya ME, and Mahmoud SH. (2026) Impact of Different Universal Adhesive Systems on the Fracture Resistance of Premolar Teeth with MOD Composite Restoration: A Literature Review. J Dent Oral Epidemiol 6(1): doi https://doi.org/10.54289/JDOE2600103
>Copyright : © 2026 Ashraf S, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Review Article | Open Access | Full Text
1Department of conservative, Faculty of Dentistry, Mansoura University, Mansoura, Egypt
2Professor, Conservative Dentistry Department, Faculty of Dentistry, Mansoura University, Mansoura, Egypt
*Corresponding author: Salah Hasab Mahmoud, Professor, Conservative Dentistry Department, Faculty of Dentistry, Mansoura University, Mansoura, Egypt
Abstract
Background: Resin composite restorations bonded to tooth structure have significantly improved contemporary restorative dentistry by supporting minimally invasive and esthetic treatment approaches. Their longevity largely depends on effective adhesion to enamel and dentin and the stability of the adhesive interface. Universal (multi-mode) adhesive systems were introduced to simplify bonding while preserving clinical performance, as they can be used in etch-and-rinse, self-etch, or selective enamel etching modes. However, concerns remain about their long-term durability and their effect on the fracture resistance of composite restored teeth.
Objectives: This review aimed to evaluate universal adhesive systems focusing on their bonding mechanisms, chemical composition particularly the role of functional monomers such as 10-methacryloyloxydecyl dihydrogen phosphate (10-MDP) and their effect on fracture resistance compared with conventional adhesive strategies.
Methods: Literature review of in vitro studies evaluating the fracture resistance of premolar teeth restored with MOD resin composite restorations using different universal adhesive systems. Analysis focused on mechanical testing protocols, including specimen preparation, loading conditions, and static fracture resistance assessment until failure with detection to the mode of failure.
Conclusion: Available evidence suggests that universal adhesive systems can influence the fracture resistance of premolar teeth restored with MOD resin composite restorations. Variations in adhesive composition and bonding strategy may affect the mechanical performance of the restored tooth structure. However, differences in mechanical testing protocols limit direct comparison among studies. Further standardized investigations are recommended to clarify the long-term mechanical behavior of these adhesive systems.
Keywords: Universal Adhesive Systems; Multi-Mode Adhesive; Etch-And-Rinse; Selfetch; Selective Enamel Etching; Fracture Resistance; Resin–Dentin Interface; Hybrid Layer; 10-MDP
Abbreviations: 4-MET: 4-Methacryloyloxyethyl Trimellitic Acid, 4-META: 4-Methacryloyloxyethyl Trimellitate Anhydride, 10-MDP: 10- Methacryloyloxydecyl Dihydrogen Phosphate, Bis-GMA: Bisphenol A-Glycidyl Methacrylate, Ca²⁺: Calcium ions, CQ: Camphorquinone, DEJ: Dentin–Enamel Junction, ER: Etch-and-Rinse, GPDM: Glycerol Phosphate Dimethacrylate, HAp: Hydroxyapatite, HEMA: 2-Hydroxyethyl Methacrylate, MOD: Mesio-OcclusoDistal, MMPs: Matrix Metalloproteinases, PAA: Polyacrylic Acid, PENTA: Dipentaerythritol Penta-Acrylate Phosphate, RMGIC: Resin-Modified Glass Ionomer Cement, SE: Self-Etch, UAs: Universal Dental Adhesives, µTBS: Microtensile Bond Strength
Search Strategy
Several international online scientific databases were systematically searched, including ResearchGate, ISI Web of Science, Scopus (SciVerse), Google Scholar, and the National Library of Medicine (MEDLINE/PubMed). The search included any published papers that evaluated the fracture resistance of teeth restored by resin composite with different universal adhesive systems. These websites were explored for studies up to 2026. The search keywords used for searching are Universal adhesive systems, Adhesion to enamel and dentin, Resin-Dentin interface and Fracture resistance.
Introduction
Adhesive dentistry has made significant progress in recent decades. With the rise of minimally invasive dentistry, this approach supports more conservative cavity preparations that depend on the effectiveness of modern enamel–dentin adhesives. The use of adhesive materials has eliminated the need for traditional mechanical retention features such as dovetails, grooves, undercuts, and sharply defined internal angles in cavity preparation [1]. As a result, modern adhesive techniques enable the preservation of a significant amount of healthy tooth structure that would otherwise be removed during conventional cavity preparation [2]. Additionally, improving the bond between restorative materials and tooth surfaces results in increased durability and success of dental restorations, resulting in greater patient comfort and a reduced risk of restoration failure [3].
Early Beginnings
In the early stages of development of dental adhesives, materials such as zinc phosphate and silicate cement were introduced to improve adhesion to tooth structures. However, these materials demonstrated insufficient bond strength and durability, limiting their effectiveness for long-term restorative applications [4]. In the 1940s, the development of improved dental adhesives led to the adoption of synthetic resins. polymethyl methacrylates resins were primarily used as fillings because of their aesthetic properties and ease of handling. However, these resins exhibited poor adhesion to tooth structures, resulting in marginal leakage and an increased risk of secondary caries. In 1955, Dr. Michael Buonocore introduced the acid-etch technique to improve bonding between resin materials and enamel [1]. This technique involved etching the enamel with phosphoric acid, which produced a microscopically rough surface that enhanced the adhesion of restorative materials by enabling better mechanical retention [5].
Dental adhesives are resin-based solutions designed to enable strong bonding between restorative materials and dental tissues. These systems contain monomers with both hydrophilic and hydrophobic components [6]. The hydrophilic portions improve wettability and promote adhesion to the moist surface of dental hard tissues, while the hydrophobic parts facilitate bonding and co-polymerization with the restorative material. In addition to monomers, dental adhesives often include curing initiators, stabilizers, inhibitors, solvents, and sometimes inorganic fillers to enhance their performance [7].
Bonding to Enamel vs Dentin
The primary goal of restorative dentistry is to achieve durable adhesion between restorative materials and tooth structures, including enamel, dentin, or both [8]. Enamel is a dry, non-vital substrate, making it an ideal surface for forming a strong and reliable adhesive bond [9]. As the enamel is a highly mineralized crystalline substance, Effective bonding to enamel is commonly achieved by etching the surface with phosphoric acid, which produces a microscopically rough texture that enhances resin retention. This etched surface is easily wetted by hydrophobic resin-based adhesives, allowing the adhesive to flow into the micro-irregularities through capillary action. As the resin polymerizes, it forms a strong micromechanical bond with enamel [10].
1. Challenges in Bonding to Dentin
1.1 Histological structure of dentin
Unlike enamel, dentin is more humid and contains a higher proportion of organic material. Dentin exhibits a complex histological architecture that varies with both anatomical location and age. It is composed of approximately 72% inorganic hydroxyapatite crystals, which are irregularly distributed within a matrix predominantly formed by collagen fibers [11]. Dentinal tubules extend from the pulp toward the dentino-enamel junction (DEJ) which is encased by a highly mineralized layer known as peritubular (intratubular) dentin and further surrounded by the less mineralized intertubular dentin [12]. The intertubular dentin primarily consists of type I collagen fibrils, with minor contributions from type III collagen, and includes a variety of non-collagenous proteins. These include proteoglycans, matrix metalloproteinases (MMPs), phosphoproteins, dentin sialoprotein, dentin glycoprotein, and other dentin-specific matrix proteins, all of which play essential roles in dentin structure and function [13].
This inherent moisture and organic composition make dentin a much more challenging substrate for adhesive bonding compared to enamel [14]. Dentinal tubules extend throughout the entire thickness of the dentin, originating from the DEJ or the cementum and continuing toward the pulp [15,16]. An increase in the diameter of dentinal tubules in deeper regions of the dentin, combined with their convergence toward the pulp chamber, leads to a significant rise in dentin permeability as cavity preparation approaches the pulp [17]. Thus, in superficial dentin, bonding occurs primarily through the intertubular dentin, whereas in deep dentin, it is predominantly intratubular due to the higher density and diameter of dentinal tubules [18].
For optimal bonding performance, any residual solvent on the dentin surface should be thoroughly evaporated by air-drying the adhesive before light-curing. If the solvent is not fully removed, it can interfere with the polymerization of resin monomers, weaken the adhesive bond, and reduce the quality of the polymer formed within the hybrid layer. Additionally, incomplete solvent evaporation can lead to the formation of voids at the adhesive interface, which may act as sites for nano leakage and ultimately reduce bond strength and mechanical durability [19,20].
Solvent evaporation can be facilitated either by allowing a waiting period between adhesive application and curing or by actively air-drying with an air syringe. Several factors influence how effectively solvents evaporate, including the type of solvent and monomer used, the distance between the air syringe and the tooth surface, the operator’s technique, and the temperature of the air, which can significantly affect the required drying time for the adhesive system [21-23].
1.2 Smear layer
Cavity preparation modifies the outermost layer of the tooth structure by creating a thin layer of cutting debris known as the smear layer, which is approximately 1.0 μm thick [24]. This layer covers the surface of the dentin and consists of pulverized hydroxyapatite and denatured collagen. Additionally, debris can penetrate and block the openings of dentinal tubules, forming what are known as smear plugs. These plugs may extend into the tubules to a depth of 1–10 µm and are continuous with the smear layer. Despite their microscopic size, the smear plugs and the smear layer can significantly affect bonding and should not be overlooked during adhesive procedures [25].
The smear layer acts as a physical barrier, significantly reducing dentinal permeability by up to 86% [26]. To ensure effective chemical bonding to the dentin surface, some degree of etching is necessary to address this obstacle, which plays a critical role in determining bond strength and long-term adhesion durability. Early generations of non-acidic adhesives were ineffective at bonding to intact dentin beneath the smear layer due to their inability to modify or penetrate it.
There are two primary strategies to overcome the challenges posed by the smear layer. The first involves its complete removal through an etch-and-rinse technique, allowing direct access to the dentin. The second approach uses self-etch adhesives that are capable of penetrating and interacting with the smear layer, incorporating it into the hybrid layer during bonding [2].
1.3 Hybrid layer
The effectiveness of the resin-dentin bond relies on the ability of the adhesive system to infiltrate the collagen matrix of dentin, which becomes exposed following acid etching. The resulting resin-dentin interdiffusion zone, known as the hybrid layer, plays a critical role in achieving micromechanical retention of dental restoration [27]. Resin monomers are unable to displace water effectively, which limits the ability of adhesive systems to fully infiltrate the demineralized collagen matrix. This incomplete penetration results in the formation of distinct zones within the hybrid layer, where micro-phase separation (referred to as micro permeability) or nano-phase separation (known as nanoleakage) can occur [28,29].
MMPs are calcium- and zinc-dependent host-derived enzymes embedded within the mineralized dentin matrix. These proteases break down the organic matrix of demineralized dentin by hydrolyzing exposed collagen fibrils beneath the hybrid layer, which significantly weakens the resin-dentin bond strength [30]. Hydrolytic degradation of the adhesive interface leads to negative clinical outcomes, including dentin hypersensitivity, marginal staining, and the risk of secondary caries, all of which reduce the longevity of dental restorations [31].
The most used monomer in dentin adhesive primers is 2-hydroxyethyl methacrylate (HEMA). Its primary role is to prepare the dental substrate for the reception of hydrophobic monomers by expanding collagen fibrils and enhancing surface wettability [32]. Owing to its low molecular weight and high hydrophilicity, HEMA significantly improves the penetration of adhesive resins into the demineralized dentin matrix. This facilitates the formation of a more uniform hybrid layer and supports the diffusion of other functional monomers, ultimately contributing to improved initial bond strength [33,34]. However, over time, the high hydrophilicity of HEMA leads to increased water sorption, which contributes to the hydrolytic degradation of the adhesive interface and compromises the long-term durability of the bond [35,36].
2. Classification of Dental Adhesives
2.1 According to chronological history
2.1.1 First generation
The first generation of dental adhesives was based on a chemical bonding method using N-phenylglycine-glycidyl methacrylate (NPG-GMA) [3]. However, the overall clinical performance of this adhesive product was limited, primarily due to high interfacial stress and thermal expansion associated with methacrylate-based composites [37].
2.1.2 Second generation
In the late 1970s, new materials were introduced to enhance the performance of first-generation dental adhesives. These adhesives primarily incorporated polymerizable phosphonates into Bisphenol-A Glycidyl Methacrylate (Bis-GMA) resins to enhance bonding with calcium ions [8]. However, they did not remove the smear layer, resulting in weak and unreliable bond strength. Due to frequent bonding failures associated with the loosely attached smear layer, this generation of adhesives is no longer in use [8]. Additionally, the presence of water in their formulation raised concerns about hydrolytic stability and long-term degradation [38].
2.1.3 Third generation
This generation emerged in mid 1980s. The clinical procedure was typically divided into two or three steps: (1) acid etching, (2) application of the primer (which, depending on the system, could be combined with the conditioner in a single bottle), and (3) application of a filler-free adhesive resin [39]. Acid etching of the dentin partially removes and/or alters the smear layer [40]. This process opened the dentin tubules, allowing the primer to be applied after the acid was thoroughly rinsed off. In most of these systems, the phosphate primer softens and modifies the smear layer; once it penetrates, it is cured to form a hardened surface. The adhesive is then applied, bonding the cured primer to the composite resin. However, bonding to smear layer–covered dentin was largely ineffective before 1990, as the resins were unable to penetrate the smear layer, and the smear layer itself was structurally weak [41].
2.1.4 Fourth generation
In the early 1990s, fourth-generation materials were the first to achieve complete removal of the smear layer [42]. One of the key features of fourth-generation bonding systems is the use of the total-etch technique [43]. This method involves etching both enamel and dentin simultaneously with 40 percent phosphoric acid for 15 to 20 seconds. After etching, the surface must remain moist, a technique called "wet bonding” to prevent the collapse of the collagen network. A hydrophilic primer is then applied, which infiltrates the exposed collagen and helps form the hybrid layer [2,44]. However, maintaining the correct level of moisture is clinically challenging; dentin that is too wet or too dry can result in suboptimal bonding [45]. Among fourth-generation etch-and-rinse systems, OptiBond FL (Kerr Corporation, Orange, CA, USA) is widely regarded as the gold standard due to its three-step protocol, filled adhesive layer, and proven long-term clinical stability [46].
Compared to other systems, fourth-generation exhibited superior microtensile bond strength, lower nanoleakage, higher degrees of conversion both overall and in situ, greater ultimate tensile strength, and reduced water sorption and solubility [47].However, the use of these systems can be quite complex and time-consuming due to the numerous bottles and application steps involved. This complexity led many dentists to seek a more simplified adhesive system.
2.1.5 Fifth generation
Manufacturers simplified adhesive systems by combining application steps, leading to the development of single-bottle systems that mix primer and adhesive. This simplification is the defining feature of fifth-generation adhesive systems [48,49]. Combining the primer and adhesive resin into a single application step necessitated the inclusion of higher concentrations of hydrophilic components in the adhesive formulations. This was essential to enhance their diffusion into the collagen network of etched dentin [50]. One commonly used monomer in simplified adhesive systems is HEMA, due to its strong hydrophilic nature, which improves dentin wettability and significantly enhances bond strength [49]. Unfortunately, the high hydrophilicity of HEMA can result in long-term drawbacks, such as increased water absorption and hydrolytic degradation of the adhesive interface [51,52]. These issues can lead to clinical problems like dentin sensitivity, marginal discoloration, and potential recurrence of caries, ultimately compromising the longevity and effectiveness of restorations [28].
2.1.6 Sixth generation
Called Self-etch adhesive systems. designed to simplify the application process by reducing the number of procedural steps. Unlike earlier generations, these systems combine etching and priming into a single step, making the hybridization process quicker, more efficient, and less prone to technique-sensitive errors [5]. Early evaluations of these new adhesive systems showed a strong bond to conditioned dentin, but the bond to enamel was less effective. This may be due to the acidic composition of sixth-generation systems, which are applied as a single solution. These solutions are difficult to keep in place, need frequent refreshing, and have a pH that isn’t strong enough to properly etch enamel [53]. To improve the bond to enamel, it is recommended to first etch the enamel with traditional phosphoric acid before applying the adhesive system.
2.1.7 Seventh generation
The latest generation of adhesive systems combines the etchant, primer, and bonding agent in a single solution, forming a one-step or “all-in-one” procedure. These self-etch adhesives are considered an attractive option by clinicians, as they simplify the bonding process by reducing the number of clinical steps compared to traditional multistep etch-and-rinse systems [54]. Cured adhesives can allow water to migrate from dentin, forming blisters at the composite–adhesive interface. These blisters, caused by adhesive permeability and trapped water, are identified as dark deposits when immersed in a silver nitrate tracer solution. This results in reduced shelf life and decreased bond strength of the adhesive [23].
2.1.8 Eighth Generation
The eighth-generation bonding agent incorporates nanosized fillers to enhance its mechanical properties and improve the stability of the adhesive interface [2]. Nanofillers with an approximate particle size of 12 nm are incorporated into these new adhesive systems. These fillers enhance the penetration of resin monomers and contribute to an increased hybrid layer thickness, which in turn improves the mechanical properties of the bonding system [38,55]. Additionally, nano-bonding agent solutions containing nano-fillers have been shown to provide better bond strength to both enamel and dentin, improved stress absorption, and extended shelf life [56]. These advanced self-etch adhesives contain acidic hydrophilic monomers that can be easily applied to moistened, etched enamel surfaces [57].
2.2 Classification according to etching approach
2.2.1 Etch-and-rinse adhesives
ER technique is performed in a multi-step process. It involves a distinct etching phase using phosphoric acid, the strongest etchant among the available methods, followed by thorough rinsing [58]. This approach effectively removes the smear layer, allowing the adhesive to penetrate the dentinal tubules without obstruction. As a result, well-formed resin tags are produced, contributing to improved micromechanical bonding [59].
The manufacturers of ER systems have developed two variations: the twostep and three-step ER systems. The main difference between these lies in how the components are package three-step ER systems keep the etchant, primer, and adhesive in separate bottles, whereas two-step ER systems combine the primer and adhesive in a single bottle, with the etchant provided separately for use prior to adhesive application. This process involves a multi-step clinical procedure starting with etching, followed by priming, and concluding with adhesive application [60].
However, the superficial dentin layer may become weakened due to the strong etching effect of the etchant used, which can also reduce the adhesive’s ability to form strong chemical bonds with the low mineral content present in the dentin hybrid layer, thereby emphasizing micromechanical adhesion [59,61]. In general, determining the optimal level of dentin moisture makes dentin bonding particularly challenging. Moreover, achieving the ideal level of "moist dentin" is difficult because enamel must be dried for bonding, yet drying the enamel without also drying the underlying dentin is hard to control [62].
2.2.2 Self-etch adhesive system
SE adhesives consist of acidic functional monomers, typically containing carboxyl or phosphate groups, that simultaneously etch and prime the tooth structure without the need for separate etching steps. Instead of removing the smear layer entirely, these adhesives modify and incorporate it [63,64]. The acidic monomers penetrate and partially demineralize the smear layer and the underlying tooth substrate, integrating the dissolved smear layer and demineralized components into the hybrid layer rather than rinsing them away [65]. Compared to etch-and-rinse adhesives, SE systems are easier to use, have a faster application process, and are less sensitive to operator technique [66].
The degree of dentin demineralization depends on the primer’s acidity, which can be classified as ultra-mild (pH ≥ 2.5), mild (pH ≈ 2), intermediately strong (pH between 1 and 2), or strong (pH < 1). Primers with lower pH values penetrate dentin more deeply. Ultra-mild and mild SE adhesives partially demineralize dentin while retaining hydroxyapatite bound to collagen, allowing for potential chemical interaction. This process also supports the formation and maintenance of nanolayering, which contributes to the durability of the adhesive bond [67,68].
Conversely, strong self-etch adhesives demineralize dentin in a way that closely resembles the action of etch-and-rinse systems. They exert an aggressive etching effect on both enamel and dentin simultaneously. In enamel, these adhesives generate an acid-etch pattern comparable to that produced by phosphoric acid. However, in dentin, unlike with phosphoric acid etching, the dissolved calcium phosphates are not rinsed away but remain trapped within the tissue [2]. These residual calcium phosphates are highly unstable and can undermine the integrity of the dentin–adhesive bond over time. Additionally, concerns have been raised about the long-term durability of strong acidic treatments on dentin due to collagen instability and enzymatic degradation result of the extreme low pH of conventional acid etchants and the ionic demineralization process.15 "Intermediately strong" self-etch adhesives produce a hybrid layer that represents a transition between "strong" and "mild" etching. This hybrid layer typically consists of a fully demineralized superficial zone and a partially demineralized underlying layer [63].
Self-etch adhesives bond to tooth by two mechanisms: chemical bond and micromechanical interlocking. Self-etch contain functional acidic monomers with carboxylic, phosphonic, or phosphate groups that can chemically bond to the remaining hydroxyapatite in dentin. This bonding improves resistance to hydrolytic breakdown, helping to preserve the restoration margins over time. Some common examples of these monomers are 10-methacryloyloxydecyl dihydrogen phosphate (10-MDP), 4-methacryloyloxyethyl trimellitic acid (4-MET), 4methacryloyloxyethyl trimellitate anhydride (4-META), 4-acryloyloxyethyl trimellitate anhydride (4-AETA), 2 methacryloyloxyethyl dihydrogen phosphate (MEP), methacryloyloxydodecylpyridinium bromide (MDPB), 11-methacryloyloxy-1, 1-undecanedicarboxylic acid (MAC-10), phenyl phosphate methacrylates, acrylic ether phosphonic acid, and other phosphoric acid esters [16,36].
Current self-etch adhesive systems are classified based on the number of clinical application steps: either one-step or two-step systems. Two-step self-etch adhesives begin with the application of a hydrophilic etching primer containing acidic monomers, which simultaneously etch and prime the tooth surface. After the solvent is evaporated, a separate hydrophobic bonding agent is applied to seal the dentin [63]. One key advantage of two-step self-etch adhesives is that their bonding effectiveness is less sensitive to the moisture level of dentin compared to etch-andrinse (ER) adhesives.
2.2.3 Resin modified glass ionomer cement
A new category of adhesives, known as resin-modified glass ionomer cement (RMGIC) adhesives, has been introduced [69]. These adhesives bond to tooth structure using the conventional conditioning approach applied to RMGIC restorative materials. This involves treating dentin with polyacrylic acid (PAA), followed by rinsing and drying [70]. Conditioning removes the smear layer except for smear plugs, partially demineralizes the dentin surface, and facilitates a chemical reaction between the glass ionomer components and hydroxyapatite in the tooth [71]. As a result, RMGIC adhesives adhere to tooth structure through two mechanisms: chemical bonding via ionic interaction between the carboxyl groups and calcium ions in the tooth, and micro-mechanical interlocking of the resin component with the conditioned dentin surface [72,73].
2.2.4 Universal Adhesive systems
Referred to as “multi-mode” or “multi-purpose” adhesives because they can be used in different application modes: as SE adhesives, ER adhesives, or in a combined approach as it is done as SE on dentin and ER on enamel or known as “selective enamel etching” [33,74]. This flexible adhesive concept supports choosing the most straightforward version of each technique by using one-step SE or two-step ER systems [75]. The key distinction between universal adhesives and traditional onestep SE adhesives lies in the inclusion of functional phosphate and/or carboxylate monomers in universal adhesives. These functional monomers can promote chemical bonding with calcium in hydroxyapatite, enhancing adhesion to tooth structure [76]. Different applications strategies shown in Figure 1.
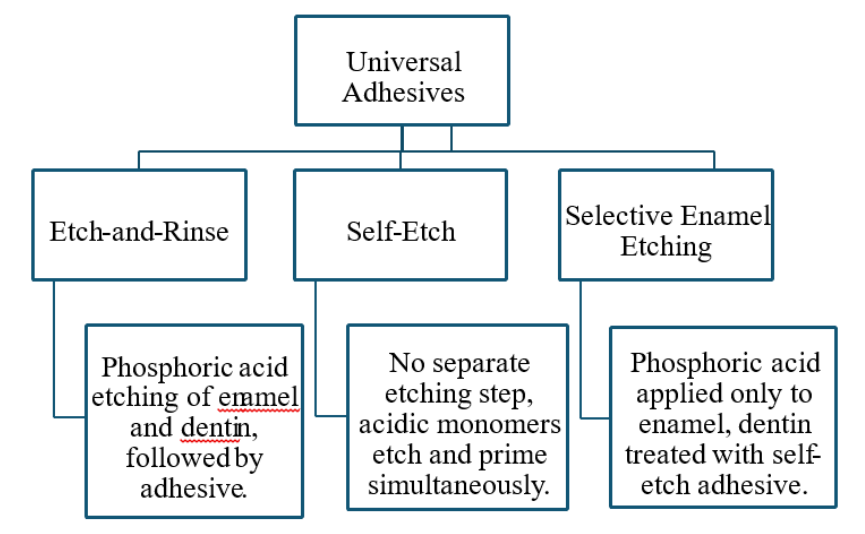
Figure 1: Different applications strategies of UAs.
Composition of Universal Adhesives
1. Functional monomers
The high bond strength to dentin is largely attributed to the chemical properties of functional monomers [77]. These monomers play a key role in determining the bonding performance of adhesive systems. Long-term adhesion is achieved through chemical interaction between the adhesive and the dentin substrate, leading to the formation of a three-dimensional collagen–resin network that establishes a stable and continuous bond [78]. UAs contain several functional monomers such as 10methacryloyloxydecyl dihydrogen phosphate (10-MDP), 4-methacryloxyethyl trimellitate anhydride (4-META), glycerol phosphate dimethacrylate (GPDM), and dipentaerythritol penta-acrylate phosphate (PENTA) [46].
10-MDP can form water-insoluble calcium salts with dentin and is therefore regarded as one of the most reliable monomers for dentin bonding [79,80]. The bifunctional nature of 10-MDP allows it to interact simultaneously with both the resin matrix and the tooth structure. It co-polymerizes with the resin while inducing controlled, pH-dependent partial demineralization of tooth hydroxyapatite (HAp), exposing calcium ions (Ca²⁺) without disrupting the collagen framework. These calcium ions subsequently form stable ionic bonds with the phosphate groups of 10MDP, resulting in the formation of self-assembled, insoluble interfacial nanolayers known as Ca–MDP nanolayering [81,82,83]. This nanolayering reinforces the mineral protected hybrid layer and contributes significantly to the long-term durability and hydrolytic stability of the adhesive interface [84].
2. Cross linking monomers
Universal adhesives feature a resin matrix composed of a strategic combination of monomers: hydrophilic (such as HEMA), hydrophobic (such as decanediol dimethacrylate [D3MA]), and moderately hydrophilic or intermediate monomers (such as bis-GMA). This tailored blend enables universal adhesives to function as an effective interface between the hydrophilic tooth substrate and the hydrophobic resin composite, ensuring reliable adhesion across a wide range of clinical conditions. Furthermore, some universal adhesives are formulated with integrated silane, which may eliminate the need for a separate silanization step when bonding to substrates like glass ceramics or resin composites. However, despite their convenience, these simplified systems have been consistently linked to inferior bond strength in laboratory studies and reduced long-term performance of restorations in clinical settings [85,86].
3. Hydrophillic monomers
HEMA is hydrophilic methacrylate monomer is widely incorporated in singlebottle adhesive systems due to its small molecular size, which facilitates resin penetration into the collagen fibril network [87]. In addition, HEMA helps reduce phase separation between hydrophilic and hydrophobic components within the adhesive formulation.
Although HEMA improves the wettability of adhesives, its high-water permeability means that high concentrations may reduce the long-term mechanical stability of the polymer due to hydrolytic degradation [88]. Consequently, HEMA-free dental adhesives were developed to improve the mechanical, chemical, and physical properties of adhesive systems [89]. Various alternative monomers have been introduced for this purpose, including dimethacrylates such as glycerol dimethacrylate (GDMA). In addition, acrylamide-based monomers have been investigated, including methacrylamide monomer, hydroxyethyl acrylamide (HEAA), and diethyl acrylamide (DEAA) [90].
4. Solvents
Water, acetone, and ethanol are the most commonly used solvents in dental adhesive systems [84]. They function to dissolve adhesive monomers, thereby reducing the viscosity of primers and resins and facilitating their penetration into etched dentin [91].
Solvents are incorporated into adhesive formulations to enhance micromechanical retention with both enamel and dentin by removing excess water from the collagen fibril network and in dissolving amphiphilic resin components, which improves surface interaction and adhesive infiltration [16].
Solvent evaporation is a critical factor influencing the bonding effectiveness of contemporary adhesive systems. The evaporation potential of different solvents varies according to their vapor pressure (mmHg), which represents the tendency of a liquid to vaporize. Solvents with higher vapor pressure exhibit greater volatility and evaporate more readily. Among the commonly used solvents in dental adhesives, acetone has higher vapor pressure than ethanol and water [92]. However, despite its high volatility, acetone does not effectively promote water evaporation because it does not form an azeotropic mixture with water [93].
Another important property of solvents is their ability to form hydrogen bonds. This allows the collagen network to re-expand after dehydration, which improves resin dispersion and enhances bonding. In addition, the polarity of the resin affects both the number of hydrogen-bonding sites and the interaction between the solvent and the polymer [94]. Newer solvents, including tert-butanol and isopropanol, have been introduced to reduce the sensitivity of adhesive systems to the level of dentin moisture. These solvents are used in adhesives such as Prime & Bond (Dentsply). Because of their relatively higher molecular weight, they reduce the likelihood of collagen fibril collapse during the bonding procedure [95].
Acidic monomers cannot dissociate without the presence of water. Conversely, an excess of water can result in phase separation, chemical degradation, decreased shelf life, and evaporation blockage during air drying. The pH range of universal adhesives is typically 1.5 to 3.2. As a result, the majority of these adhesives fall into one of three categories: mild (pH ~ 2), ultra-mild (pH >2.5), or intermediately strong (pH < 2) [23]. The complicated formulation of simplified adhesives and their high solvent concentration, which can prevent full solvent volatilization and subsequently result in worse adhesive polymerization [96].
5. Photoiniators and fillers
Photoinitiators and fillers are important components of dental adhesive systems. Photoinitiators initiate the polymerization process when the adhesive is exposed to visible light. The most commonly used photoinitiator in dental resins is camphorquinone (CQ), which reacts with a tertiary amine co-initiator to generate free radicals that convert monomers into a polymer network [97]. In addition, some adhesive formulations incorporate inorganic fillers, such as silica or glass nanoparticles, to improve the viscosity, mechanical properties, and stability of the adhesive layer [46].
Despite the growing popularity of universal adhesives, information regarding their bonding mechanisms and film-forming characteristics remains limited [98-100]. with most of the data coming from the manufacturers Given that the continuous search for clinical step reduction and enhanced technique sensitivity led to the appearance of these materials as a new trend in dental adhesion [101]. Manufacturers often state that universal adhesives work with resin-based adhesives that are selfcure, light-cure, and dual-cure, and that they can be used with both direct and indirect restorations [102].
Fracture Resistance
Fracture toughness is a mechanical property that describes the resistance of a brittle material to crack propagation under an applied load, thereby reflecting its damage tolerance [103]. The values of fracture toughness depend on the physical properties and chemical composition of each component within the restorative material. A material with high fracture toughness can better resist crack initiation and propagation. Consequently, fracture toughness and flexural strength are important factors in determining the longevity of dental materials [104,103].
Typically, the fracture toughness values correspond to rather large standard deviations (>25%). One would anticipate significantly lower standard deviation results and improved material discrimination given that fracture toughness is an intrinsic feature. Nonetheless, given the biological composition of dentin and enamel, compositional differences between specimens may influence and account for these variances in hardness levels. The variability may also be influenced by additional elements, such as the depth and orientation of the enamel and dentin substrates. Some of the heterogeneity may also be caused by the often intricate sample preparation methods [105].
Fracture mechanics offers an appropriate framework for evaluating dental adhesives. However, measuring fracture toughness at interfaces involving two or more materials is more complex than testing single-component systems. Most studies on dental adhesives have overlooked the intricate stress conditions within the transitional zones of adhesive joints [105].
It is known that cycling fatigue loading more accurately replicates clinical conditions than static loading because it produces cyclic forces that are comparable to typical masticatory forces. Fracture resistance and fatigue failure might be assessed utilizing a universal testing machine [106,107]. As an appropriate balance between the traditional load-to-fracture test and the more complex and time consuming fatigue testing, the accelerated fatigue procedure was devised. Because of the stresses in the posterior region during mastication range from 500 - 900 N, the accelerated fatigue test was only conducted up to 1000 N. Although this test has been done with heavier loads in certain studies, it would not have been practical in this case [108,109].
Saitamon et al. [110] evaluated the fracture resistance of MOD resin composite restorations using different adhesive strategies. They concluded that Scotchbond Multi-Purpose demonstrated the highest fracture resistance values. Selective enamel etching with Single Bond Universal improved and stabilized fracture resistance compared to the self-etch approach, while thermal aging adversely affected performance primarily in the etch-and-rinse mode of the universal adhesive.
Coelho-de-Souza FH et al. [111] evaluated the effect of different adhesive systems on the fracture resistance of premolars restored with composite resin. The results showed that the use of a total-etch (etch-and-rinse) adhesive system significantly increased fracture resistance compared to self-etch approaches, particularly when combined with appropriate margin preparation.
Al-Jedai et al. [112] conducted a comparative study evaluating the interface fracture behavior of a fourth-generation three-step adhesive and a seventh-generation universal adhesive. The results showed that the multi-step adhesive exhibited superior interfacial fracture resistance and integrity compared with the universal adhesive, which demonstrated greater technique sensitivity despite its simplified application.
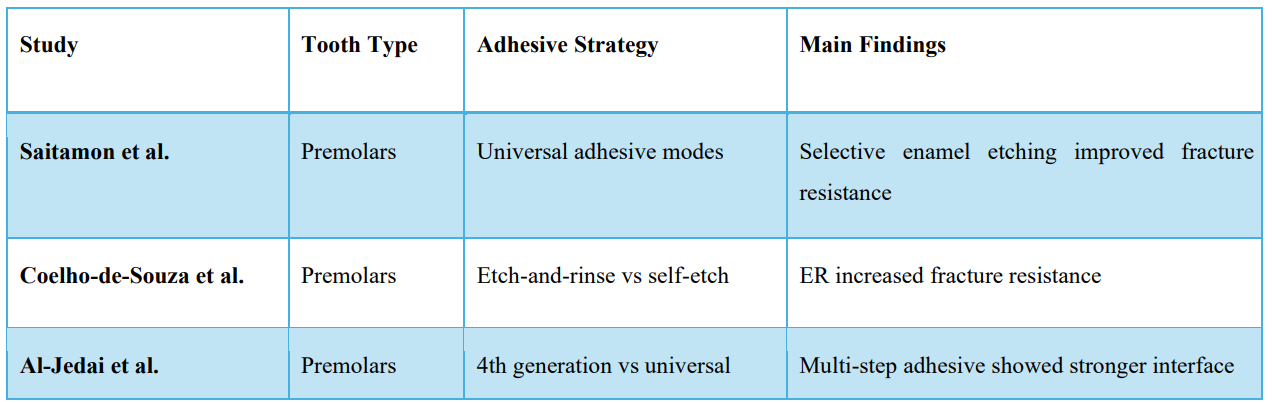
Table 1: Main findings of previous studies
References
- Pinna R., Usai P., Filigheddu E., Garcia-Godoy F., Milia E. The role of adhesive materials and oral biofilm in the failure of adhesive resin restorations. Am J Dent. 2017;30:285–292. [PubMed.]
- Sofan E., Sofan A., Palaia G., Tenore G., Romeo U., Migliau G. Classification review of dental adhesive systems: from the IV generation to the universal type. Ann Stomatol (Roma). 2017;8:1–17. [PubMed.]
- Waad Khalid Alomran., Mohammed Zahedul Islam Nizami., Hockin HKXu., andJirun Sun. Evolution of Dental Resin Adhesives—A Comprehensive Review. J Funct Biomater. 2025;16:104. [Ref.]
- Mount GJ., Hume WR., Ngo HC., Wolff MS. Preservation and restoration of tooth structure: John Wiley & Sons. 2016;1998-579. [Ref.]
- Van Meerbeek B., Yoshihara K., Yoshida Y., Mine A., De Munck J., Van Landuyt KL. State of the art of self-etch adhesives. Dent Mater. 2011;27:17–28. [PubMed.]
- Perdigao J. New developments in dental adhesion. Dent Clin North Am. 2007;51:333–357. [PubMed.]
- Reis A., Loguercio AD., Manso AP., Grande RH., Schiltz-Taing M., Suh B., et al. Microtensile bond strengths for six 2-step and two 1-step self-etch adhesive systems to enamel and dentin. Am J Dent. 2013;26:44–50. [PubMed] [PubMed.]
- Van Meerbeek B., Yoshihara K., Van Landuyt K., Yoshida Y., Peumans M. From Buonocore's Pioneering Acid-Etch Technique to Self-Adhering Restoratives. A Status Perspective of Rapidly Advancing Dental Adhesive Technology. J Adhes Dent. 2020;22:7–34. [PubMed.]
- Perdigao J. Current perspectives on dental adhesion: (1) Dentin adhesion - not there yet. Jpn Dent Sci Rev. 2020;56:190–207. [PubMed] [PubMed.]
- Bracher L., and Özcan M. Adhesion of resin composite to enamel and dentin: a methodological assessment. J Adhes Sci Technol. 2018;32:258–271. [Ref.]
- Chen L., Suh BI. Effect of hydrophilicity on the compatibility between a dualcuring resin cement and one-bottle simplified adhesives. J Adhes Dent. 2013;15:325–331. [PubMed.]
- De Munck J., Mine A., Poitevin A., Van Ende A., Cardoso MV., Van Landuyt KL., et al. Meta-analytical review of parameters involved in dentin bonding. J Dent Res. 2012;91:351–357. [PubMed.]
- Benjwal S., Goswami M., Saxena A., Kurien RS., Mushtaq A. Comparative evaluation of bond strength of three self-adhering flowable composites to sound and demineralized enamel–An in vitro study. J Glob Oral Health. 2021;4:3. [Ref.]
- Perdigao J. Dentin bonding—Variables related to the clinical situation and the substrate treatment. Dent Mater. 2010;26:e24–e37. [PubMed.]
- Ikemura K., Kadoma Y., Endo T. A review of the developments of self-etching primers and adhesives -Effects of acidic adhesive monomers and polymerization initiators on bonding to ground, smear layer-covered teeth. Dent Mater J. 2011;30:769–789. [PubMed.]
- Van Landuyt KL., Snauwaert J., De Munck J., Peumans M., Yoshida Y., Poitevin A., et al. Systematic review of the chemical composition of contemporary dental adhesives. Biomaterials. 2007;28:3757–3785. [PubMed.]
- Perdigao J. Dentin bonding-variables related to the clinical situation and the substrate treatment. Dent Mater. 2010;26:e24–37. [PubMed.]
- Masarwa N., Mohamed A., Abou-Rabii I., Zaghlan RA., Steier L. Longevity of self-etch dentin bonding adhesives compared to etch-and-rinse dentin bonding adhesives: a systematic review. J Evid Based Dent Pract. 2016;16:96–106. [PubMed.]
- Chimeli TBC., D'ALPINO PHP., Pereira PN., Hilgert LA., Di Hipolito V., Garcia FCP. Effects of solvent evaporation on water sorption/solubility and nanoleakage of adhesive systems. J Appl Oral Sci. 2014;22:294–301. [PubMed.]
- Ferreira JC., Pires PT., Azevedo AF., Oliveira SA., Melo PR., Silva MJ. Influence of solvents and composition of etch-and-rinse and self-etch adhesive systems on the nanoleakage within the hybrid layer. J Contemp Dent Pract. 2013;14:691. [PubMed.]
- Hardan L., Bourgi R., Cuevas-Suárez CE., Zarow M., Kharouf N., Mancino D., et al. The bond strength and antibacterial activity of the universal dentin bonding system: a systematic review and meta-analysis. Microorganisms. 2021;9:1230. [PubMed.]
- Hardan L., Daood U., Bourgi R., Cuevas-Suarez CE., Devoto W., Zarow M., et al. Effect of collagen crosslinkers on dentin bond strength of adhesive systems: a systematic review and meta-analysis. Cells. 2022;11:2417. [PubMed.]
- Nagarkar S., Theis-Mahon N., Perdigao J. Universal dental adhesives: Current status., laboratory testing, and clinical performance. J Biomed Mater Res B Appl Biomater. 2019;107:2121–2131. [PubMed.]
- Scholz KJ., Bittner A., Cieplik F., Hiller KA., Schmalz G., Buchalla W., et al. Micromorphology of the Adhesive Interface of Self-Adhesive Resin Cements to Enamel and Dentin. Materials (Basel). 2021;14. [PubMed.]
- McLeod ME., Price RB., Felix CM. Effect of configuration factor on shear bond strengths of self-etch adhesive systems to ground enamel and dentin. Oper Dent. 2010;35:84–93. [PubMed.]
- Dündar M., Ozcan M., Cömlekoglu ME., Sen BH. Nanoleakage inhibition within hybrid layer using new protective chemicals and their effect on adhesion. J Dent Res. 2011;90:93–98. [PubMed.]
- De Munck J., Mine A., Van den Steen PE., Van Landuyt KL., Poitevin A., Opdenakker G., et al. Enzymatic degradation of adhesive-dentin interfaces produced by mild self-etch adhesives. Eur J Oral Sci. 2010;118:494–501. [PubMed.]
- Betancourt DE., Baldion PA., Castellanos JE. Resin-Dentin Bonding Interface: Mechanisms of Degradation and Strategies for Stabilization of the Hybrid Layer. Int J Biomater. 2019;2019:5268342. [PubMed.]
- Cuevas-Suárez CE., da Rosa WLO., Lund RG., da Silva AF., Piva E. Bonding Performance of Universal Adhesives: An Updated Systematic Review and MetaAnalysis. J Adhes Dent. 2019;21:7–26. [PubMed.]
- Breschi L., Mazzoni A., Ruggeri A., Cadenaro M., Di Lenarda R., et al. Dental adhesion review: aging and stability of the bonded interface. Dent Mater. 2008;24:90–101. [PubMed.]
- Kermanshahi S., Santerre JP., Cvitkovitch DG., Finer Y. Biodegradation of resin-dentin interfaces increases bacterial microleakage. J Dent Res. 2010;89:996–1001. [PubMed.]
- Pupo YM., Michél MD., Gomes OM., Lepienski CM., Gomes JC. Effect of the regional variability of dentinal substrate and modes of application of adhesive systems on the mechanical properties of the adhesive layer. J Conserv Dent. 2012;15:132–136. [PubMed.]
- Hanabusa M., Mine A., Kuboki T., Momoi Y., Van Ende A., Van Meerbeek B., et al. Bonding effectiveness of a new 'multi-mode' adhesive to enamel and dentine. J Dent. 2012;40:475–484. [PubMed.]
- Pimentel de Oliveira R., de Paula BL., Ribeiro ME., Alves E., Costi HT., Silva C. Evaluation of the Bond Strength of Self-Etching Adhesive Systems Containing HEMA and 10-MDP Monomers: Bond Strength of Adhesives Containing HEMA and 10-MDP. Int J Dent. 2022;2022:5756649. [PubMed.]
- Chowdhury A., Alam A., Yamauti M., Álvarez Lloret P., Saikaew P., et al. Characterization of an Experimental Two-Step Self-Etch Adhesive's Bonding Performance and Resin-Dentin Interfacial Properties. Polymers (Basel). 2021;13. [PubMed.]
- Manuja N., Nagpal R., Pandit IK. Dental adhesion: mechanism., techniques and durability. J Clin Pediatr Dent. 2012;36:223–234. [PubMed.]
- Ferracane JL. Resin composite--state of the art. Dent Mater. 2011;27:29–38. [PubMed.]
- Breschi L., Maravic T., Cunha SR., Comba A., Cadenaro M., et al. Dentin bonding systems: From dentin collagen structure to bond preservation and clinical applications. Dent Mater. 2018;34:78–96. [PubMed.]
- Tjaderhane L. Dentin bonding: can we make it last? Oper Dent. 2015;40:4–18. [PubMed.]
- Saikaew P., Sattabanasuk V., Harnirattisai C., Chowdhury A., Carvalho R., Sano H. Role of the smear layer in adhesive dentistry and the clinical applications to improve bonding performance. Jpn Dent Sci Rev. 2022;58:59–66. [PubMed.]
- Alomran WK., Nizami MZI., Xu HHK., Sun J. Evolution of Dental Resin Adhesives-A Comprehensive Review. J Funct Biomater. 2025;16. [PubMed.]
- Calinoiu SG., Bicleșanu C., Florescu A., Stoia DI., Dumitru C., et al. Comparative Study on Interface Fracture of 4th Generation 3-Steps Adhesive and 7th Generation Universal Adhesive. Materials (Basel). 2023;16 (17):5834. [PubMed.]
- Spencer P., Ye Q., Park J., Topp EM., Misra A., Marangos O., et al. Adhesive/Dentin interface: the weak link in the composite restoration. Ann Biomed Eng. 2010;38:1989–2003. [PubMed.]
- Luque-Martinez IV., Perdigao J., Muñoz MA., Sezinando A., Reis A., Loguercio AD. Effects of solvent evaporation time on immediate adhesive properties of universal adhesives to dentin. Dent Mater. 2014;30:1126–1135. [PubMed.]
- Ahmed B., El-Maksoud OA., Wafaie RA., Mahmoud SH. Effect of short dentin etching and water storage on bonding of resin composite to dentin with universal and two-step self-etch adhesive systems. Sci Rep. 2025;15:43817. [Ref.]
- Bourgi R., Kharouf N., Cuevas-Suárez CE., Lukomska-Szymanska M., et al. A Literature Review of Adhesive Systems in Dentistry: Key Components and Their Clinical Applications. Appl Sci. 2024;14:8111. [Ref.]
- Loguercio A., Luque-Martinez I., Muñoz M., Szesz A., Cuadros-Sánchez J., Reis A. A comprehensive laboratory screening of three-step etch-and-rinse adhesives. Oper Dent. 2014;39:652–662. [PubMed.]
- Ayar MK. Bond durability of contemporary adhesive systems to pulp chamber dentin. Acta Biomater Odontol Scand. 2015;1:76–80. [PubMed.]
- Miyazaki M., Tsujimoto A., Tsubota K., Takamizawa T., Kurokawa H., Platt JA. Important compositional characteristics in the clinical use of adhesive systems. J Oral Sci. 2014;56:1–9. [PubMed.]
- Ye Q., Park J., Parthasarathy R., Pamatmat F., Misra A., Laurence JS., et al. Quantitative analysis of aqueous phase composition of model dentin adhesives experiencing phase separation. J Biomed Mater Res B Appl Biomater. 2012;100:1086–1092. [PubMed.]
- da Silva TSP., de Castro RF., Magno MB., Maia LC., Silva ESMHDJ. Do HEMA-free adhesive systems have better clinical performance than HEMAcontaining systems in noncarious cervical lesions? A systematic review and meta-analysis. J Dent. 2018;74:1–14. [PubMed.]
- Scotti N., Comba A., Gambino A., Manzon E., Breschi L., Paolino D., et al. Influence of operator experience on non-carious cervical lesion restorations: Clinical evaluation with different adhesive systems. Am J Dent. 2016;29:33–38. [PubMed.]
- Tran XV., Tran KQ. Microleakage and characteristics of resin-tooth tissues interface of a self-etch and an etch-and-rinse adhesive systems. Restor Dent Endod. 2021;46:e30. [PubMed.]
- Alla R. Composite Resins (Tooth colored restorative materials) in. Dental Materials Science, Jaypee Brothers Medical Publishers (P) Ltd, New Delhi, India. 2013:132–148. [Ref.]
- Joseph P., Yadav C., Satheesh K., Rahna R. Comparative evaluation of the bonding efficacy of sixth, seventh and eighth generation bonding agents: an in vitro study. Int Res J Pharm. 2013;4:143–147. [PubMed.]
- M Kurami N., S Javadi N. Sealant Microleakage in saliva-contaminated enamel: comparison between three adhesive system. J Dent Sch Shahid Beheshti Univ Med Sci. 2010;27:197–204. [Ref.]
- Coelho A., Canta JP., Martins JNR., Arantes-Oliveira S., Marques PF. Influence of self-etch all-in-one adhesives on fissure sealant shear bond strength under contaminated enamel conditions. Eur Arch Paediatr Dent. 2019;20:15–22. [PubMed.]
- Alanazi AM., Khan AA., Siddiqui YTS., Leemani MJ., Shabbir T., et al. Photoactivated rose bengal‐doped TiO2 nanoparticles modified fifth‐generation adhesive on the survival rate of Streptococcus mutants and mechanical properties of tooth‐colored restorative material to carious dentin. Microsc Res Tech. 2024;87:2943–2953. [PubMed.]
- Alam A., Yamauti M., Chowdhury A., Wang X., Álvarez-Lloret P., et al. Evaluating the advancements in a recently introduced universal adhesive compared to its predecessor. J Dent Sci. 2024;19:1609–1619. [PubMed.]
- Kawazu M., Takamizawa T., Hirokane E., Tsujimoto A., Tamura T., Barkmeier WW., et al. Comparison of dentin bond durability of a universal adhesive and two etch-and-rinse adhesive systems. Clin Oral Investig. 2020;24:2889–2897. [PubMed.]
- Wang HM., Li KX., Tian ZL., Zhu YL., Liu XY., Yang SH., et al. New Monomer Capable of Dual Chemical Binding with Dentin to Improve Bonding Durability. J Dent Res. 2024;103:820–829. [PubMed.]
- Perdigao J., Reis A., Loguercio AD. Dentin adhesion and MMPs: a comprehensive review. J Esthet Restor Dent. 2013;25:219–241. [PubMed.]
- Giannini M., Makishi P., Ayres AP., Vermelho PM., Fronza BM., Nikaido T., et al. Self-etch adhesive systems: a literature review. Braz Dent J. 2015;26:3–10. [PubMed.]
- Perdigao J., Araujo E., Ramos RQ., Gomes G., Pizzolotto L. Adhesive dentistry: Current concepts and clinical considerations. J Esthet Restor Dent. 2021;33:51–68. [PubMed.]
- Peumans M., De Munck J., Mine A., Van Meerbeek B. Clinical effectiveness of contemporary adhesives for the restoration of non-carious cervical lesions. A systematic review. Dent Mater. 2014;30:1089–1103. [PubMed.]
- Gutiérrez MF., Bermudez J., Dávila-Sánchez A., Alegría-Acevedo LF., MéndezBauer L., Hernández M., et al. Zinc oxide and copper nanoparticles addition in universal adhesive systems improve interface stability on caries-affected dentin. J Mech Behav Biomed Mater. 2019;100:103366. [PubMed.]
- Yoshida Y., Yoshihara K., Nagaoka N., Hayakawa S., et al. Self-assembled Nano-layering at the Adhesive interface. J Dent Res. 2012;91:376–381. [PubMed.]
- Mahn E., Rousson V., Heintze S. Meta-Analysis of the Influence of Bonding Parameters on the Clinical Outcome of Tooth-colored Cervical Restorations. J Adhes Dent. 2015;17:391–403. [PubMed.]
- Sidhu SK., Nicholson JW. A Review of Glass-Ionomer Cements for Clinical Dentistry. J Funct Biomater. 2016;7. [PubMed.]
- Moshaverinia M., Navas A., Jahedmanesh N., Shah KC., Moshaverinia A., Ansari S. Comparative evaluation of the physical properties of a reinforced glass ionomer dental restorative material. J Prosthet Dent. 2019;122:154–159. [PubMed.]
- Cardoso MV., Delmé KI., Mine A., Neves Ade A., Coutinho E., et al. Towards a better understanding of the adhesion mechanism of resin-modified glass-ionomers by bonding to differently prepared dentin. J Dent. 2010;38:921–929. [PubMed.]
- Park EY., Kang S. Current aspects and prospects of glass ionomer cements for clinical dentistry. Yeungnam Univ J Med. 2020;37:169–178. [PubMed.]
- Perdigao J., Sezinando A., Monteiro PC. Laboratory bonding ability of a multipurpose dentin adhesive. Am J Dent. 2012;25:153–158. [PubMed.]
- Muñoz MA., Luque I., Hass V., Reis A., Loguercio AD., Bombarda NH. Immediate bonding properties of universal adhesives to dentine. J Dent. 2013;41:404–411. [PubMed.]
- Yoshida Y., Yoshihara K., Nagaoka N., Hayakawa S., et al. Self-assembled Nano-layering at the Adhesive interface. J Dent Res. 2012;91:376–381. [PubMed.]
- Yoshihara K., Yoshida Y., Nagaoka N., Hayakawa S., Okihara T., De Munck J., et al. Adhesive interfacial interaction affected by different carbon-chain monomers. Dent Mater. 2013;29:888–897. [PubMed.]
- Wang X., Wang C., Zhang L., Zhang Z., Fu B., Hannig M. Influence of priming time and primer’s concentrations on bovine enamel bond strengths. J Adhes Sci Technol. 2013;27:2558–2570. [Ref.]
- Prasansuttiporn T., Nakajima M., Foxton RM., Tagami J. Scrubbing effect of self-etching adhesives on bond strength to NaOCl-treated dentin. J Adhes Dent. 2012;14:121–127. [PubMed.]
- Carrilho E., Cardoso M., Marques Paula A., et al. 10-MDP Based Dental Adhesives: Adhesive Interface Characterization and Adhesive Stability-A Systematic Review. Materials (Basel). 2019;12. [PubMed.]
- Wang R., Shi Y., Li T., Pan Y., Cui Y., Xia W. Adhesive interfacial characteristics and the related bonding performance of four self-etching adhesives with different functional monomers applied to dentin. J Dent. 2017;62:72–80. [PubMed.]
- Fehrenbach J., Isolan CP., Münchow EA. Is the presence of 10-MDP associated to higher bonding performance for self-etching adhesive systems? A meta-analysis of in vitro studies. Dent Mater. 2021;37:1463–1485. [PubMed.]
- Han F., Jin X., Yuan X., Bai Z., Wang Q., Xie H. Interactions of two phosphate ester monomers with hydroxyapatite and collagen fibers and their contributions to dentine bond performance. J Dent. 2022;122:104159. [PubMed.]
- Yaguchi T. Layering mechanism of MDP-Ca salt produced in demineralization of enamel and dentin apatite. Dent Mater. 2017;33:23–32. [PubMed.]
- Fujita Nakajima K., Nikaido T., Arita A., Hirayama S., Nishiyama N. Demineralization capacity of commercial 10-methacryloyloxydecyl dihydrogen phosphate-based all-in-one adhesive. Dent Mater. 2018;34:1555–1565. [PubMed.]
- Munoz MA., Sezinando A., Luque-Martinez I., Szesz AL., Reis A., Loguercio AD., et al. Influence of a hydrophobic resin coating on the bonding efficacy of three universal adhesives. J Dent. 2014;42:595–602. [PubMed.]
- Tuncer D., Yazici AR., Özgünaltay G., Dayangac B. Clinical evaluation of different adhesives used in the restoration of non-carious cervical lesions: 24-month results. Aust Dent J. 2013;58:94–100. [PubMed.]
- Alex G. Universal adhesives: the next evolution in adhesive dentistry? Compend Contin Educ Dent. 2015;36:15–26;quiz 28, 40. [PubMed.]
- Reichl FX., Lohle J., Seiss M., Furche S., Shehata MM., Hickel R., et al. Elution of TEGDMA and HEMA from polymerized resin-based bonding systems. Dent Mater. 2012;28:1120–1125. [PubMed.]
- Münchow E., Zanchi C., Ogliari F., Silva M., De Oliveira I., Piva E. Replacing HEMA with Alternative Dimethacrylates in Dental Adhesive Systems: Evaluation of Polymerization Kinetics and Physicochemical Properties. J Adhes Dent. 2014;16. [PubMed.]
- Ahmed MH., Yoshihara K., Yao C., Okazaki Y., Van Landuyt K., et al. Multiparameter evaluation of acrylamide HEMA alternative monomers in 2-step adhesives. Dent Mater. 2021;37:30–47. [PubMed.]
- Lins R., Sebold M., Magno MB., Maia LC., Martins L., Giannini M. Does the Type of Solvent in Dental Adhesives Influence the Clinical Performance of Composite Restorations Placed in Noncarious Cervical Lesions? A Systematic Review and Meta-analysis. Oper Dent. 2020;45:E237–e254. [PubMed.]
- Carvalho CN., Lanza MDS., Dourado LG., Carvalho EM., Bauer J. Impact of Solvent Evaporation and Curing Protocol on Degree of Conversion of Etch-andRinse and Multimode Adhesives Systems. Int J Dent. 2019;2019:5496784. [PubMed.]
- Chowdhury A., Saikaew P., Alam A., Sun J., Carvalho RM., Sano H. Effects of Double Application of Contemporary Self-Etch Adhesives on Their Bonding Performance to Dentin with Clinically Relevant Smear Layers. J Adhes Dent. 2019;21:59–66. [PubMed.]
- Yiu CK., Pashley EL., Hiraishi N., King NM., Goracci C., Ferrari M., et al. Solvent and water retention in dental adhesive blends after evaporation. Biomaterials. 2005;26:6863–6872. [PubMed.]
- Dressano D., Salvador MV., Oliveira MT., Marchi GM., Fronza BM., Hadis M., et al. Chemistry of novel and contemporary resin-based dental adhesives. J Mech Behav Biomed Mater. 2020;110:103875. [PubMed.]
- Yoshihara K., Nagaoka N., Sonoda A., Maruo Y., Makita Y., Okihara T., et al. Effectiveness and stability of silane coupling agent incorporated in 'universal' adhesives. Dent Mater. 2016;32:1218–1225. [PubMed.]
- Kowalska A., Sokolowski J., Bociong K. The Photo initiators Used in Resin Based Dental Composite-A Review and Future Perspectives. Polymers (Basel). 2021;13. [PubMed.]
- Cardenas AM., Siqueira F., Rocha J., Szesz AL., Anwar M., et al. Influence of Conditioning Time of Universal Adhesives on Adhesive Properties and Enamel-Etching Pattern. Oper Dent. 2016;41:481–490. [PubMed.]
- Kusakabe S., Rawls HR., Hotta M. Relationship between thin-film bond strength as measured by a scratch test., and indentation hardness for bonding agents. Dent Mater. 2016;32:e55–62. [PubMed.]
- Loguercio AD., Muñoz MA., Luque-Martinez I., Hass V., Reis A., Perdigao J. Does active application of universal adhesives to enamel in self-etch mode improve their performance? J Dent. 2015;43:1060–1070. [PubMed.]
- Jang JH., Lee MG., Woo SU., Lee CO., Yi JK., Kim DS. Comparative study of the dentin bond strength of a new universal adhesive. Dent Mater J. 2016;35:606–612. [PubMed.]
- Papadogiannis D., Dimitriadi M., Zafiropoulou M., Gaintantzopoulou MD., Eliades G. Universal Adhesives: Setting Characteristics and Reactivity with Dentin. Materials (Basel). 2019;12. [PubMed.]
- Ornaghi BP., Meier MM., Lohbauer U., Braga RR. Fracture toughness and cyclic fatigue resistance of resin composites with different filler size distributions. Dent Mater. 2014;30:742–751. [PubMed.]
- Heintze SD., Ilie N., Hickel R., Reis A., Loguercio A., Rousson V. Laboratory mechanical parameters of composite resins and their relation to fractures and wear in clinical trials—A systematic review. Dent Mater. 2017;33:e101–e114. [PubMed.]
- Soderholm K-J. Review of the fracture toughness approach. Dent Mater. 2010;26:e63–e77. [PubMed.]
- Lazari PC., de Carvalho MA., Cury AADB., Magne P. Survival of extensively damaged endodontically treated incisors restored with different types of posts-andcore foundation restoration material. J Prosthet Dent. 2018;119:769–776. [PubMed.]
- Magne P., Lazari P., Carvalho M., Johnson T., Del Bel Cury A. Ferrule-effect dominates over use of a fiber post when restoring endodontically treated incisors: an in vitro study. Oper Dent. 2017;42:396–406. [PubMed.]
- Arola D. Fatigue testing of biomaterials and their interfaces. Dent Mater. 2017;33:367–381. [PubMed.]
- Koc D., Dogan A., Bek B. Bite force and influential factors on bite force measurements: a literature review. Eur J Dent. 2010;4:223–232. [PubMed.]
- Saitamon P., Pravalpruekskul S., Vongphan N., Harnirattisai C., Sattabanasuk V. Universal Adhesive Application Modes Differentially Affect the Fracture Resistance of Resin Composite Restored Teeth. Oper Dent. 2024;49:200–209. [PubMed.]
- Coelho-de-Souza FH., Rocha Ada C., Rubini A., Klein-Júnior CA., Demarco FF. Influence of adhesive system and bevel preparation on fracture strength of teeth restored with composite resin. Braz Dent J. 2010;21:327–331. [PubMed.]
- Calinoiu ȘG., Bicleșanu C., Florescu A., Stoia DI., Dumitru C., et al. Comparative Study on Interface Fracture of 4th Generation 3-Steps Adhesive and 7th Generation Universal Adhesive. Materials (Basel). 2023;16:5834. [PubMed.]