>Corresponding Author : Salah Hasab Mahmoud
>Article Type : Review Article
>Volume : 6 | Issue : 1
>Received Date : 19 Feb, 2026
>Accepted Date : 06 March, 2026
>Published Date : 11 March, 2026
>DOI : https://doi.org/10.54289/JDOE2600102
>Citation : Diwan SY, Ismail HS, and Mahmoud SH. (2026) Antibacterial Properties and Fluoride Release of Contemporary Bioactive Restorative Materials. J Dent Oral Epidemiol 6(1): doi https://doi.org/10.54289/JDOE2600102
>Copyright : © 2026 Diwan SY, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Review Article | Open Access | Full Text
1MSc Candidate in operative dentistry, Faculty of dentistry, Mansoura university, Mansoura, Egypt
2Lecturer, Conservative Dentistry Department, Faculty of Dentistry, Mansoura University, Mansoura, Egypt
3Professor, Conservative Dentistry Department, Faculty of Dentistry, Mansoura University, Mansoura, Egypt
*Corresponding author: Salah Hasab Mahmoud, Professor, Conservative Dentistry Department, Faculty of Dentistry, Mansoura University, Mansoura, Egypt
Abstract
Background: Dental caries is a highly prevalent oral disease globally, leading to significant restorative challenges. Bioactive dental materials have emerged as advanced restorative solutions combining mechanical strength, antibacterial properties and ion releasing ability (especially fluoride). These materials, including glass ionomer cements (GICs), resin-modified GICs (RMGIs), high-viscosity GICs (HVGICs), compomers, giomers, alkasite composites, and bioactive composites (ACTIVA), aim to restore teeth while preventing secondary caries. This review explores their chemistry, clinical applications, antibacterial activity, ion release, and anticariogenic potential, highlighting their role in minimally invasive dentistry.
Objectives: To evaluate the effectiveness of bioactive restorative materials in secondary caries prevention, to assess the antibacterial activity of these materials against cariogenic bacterial, to compare fluoride and ion release among different restorative materials, and to summarize their mechanical properties and clinical applications.
Aim: To provide a comprehensive understanding of the bioactive properties of contemporary restorative materials and their impact on caries management, including antibacterial effect, fluoride release, and restoration longevity.
Methods: Literature review of in vitro and clinical studies on bioactive restorative materials. Analysis of material composition, antibacterial activity, fluoride/ion release, and mechanical properties. Comparison of conventional and modified materials (GIC, RMGI, HVGIC, Compomer, Giomer, Alkasite, ACTIVA) based on clinical applications and evidence of anticariogenicity. Review of methods used to assess antibacterial activity (agar diffusion, bioluminescence, time-kill kinetics, co-culture) and fluoride release (spectrophotometry, ion chromatography, mass spectrometry, ion-selective electrodes).
Results: GICs: Moderate antibacterial activity, high fluoride release, limited mechanical strength. RMGICs: Moderate antibacterial activity, moderate fluoride release, improved mechanical strength. HVGICs: Strong antibacterial activity, good fluoride release, high mechanical strength. Compomers: Good aesthetics, limited fluoride release, moderate mechanical strength. Giomer: superior aesthetics, minimal antibacterial activity, low fluoride release, moderate mechanical strength. Alkasite composites: Moderate antibacterial activity, moderate fluoride/ion release, high mechanical strength. Modified resin composite: Strong antibacterial effect, bioactive fluoride and ion release, high mechanical strength.
Conclusion: Bioactive restorative materials represent a significant advancement in restorative dentistry. They integrate mechanical durability with biological activity, providing antibacterial activity, ion release, and remineralization that contribute to secondary caries prevention. While traditional GICs excel in fluoride release, modern materials like HVGICs, Alkasite composites, and modified resin composite combine aesthetics, bioactivity, and strength. Further clinical research is needed to evaluate their interaction with oral biofilm, long-term effectiveness, and ion release dynamics, to optimize restorative outcomes.
Keywords: Bioactive restorative materials, glass ionomer, antibacterial activity, fluoride release, remineralization, minimally invasive dentistry
Abbreviations: GIC: Glass Ionomer Cement, RMGI: Resin-Modified Glass Ionomer, HVGIC: High Viscosity Glass Ionomer Cement, FAS: Fluoroaluminosilicate, PAA: Polyacrylic Acid, HEMA: 2-Hydroxyethyl Methacrylate, Bis-GMA: Bisphenol A-Glycidyl Methacrylate, TEGDMA: Triethylene Glycol Dimethacrylate, UDMA: Urethane Dimethacrylate, PRG: Pre-Reacted Glass, F-PRG: Full Reaction Type Pre-Reacted Glass, S-PRG: Surface Reaction Type Pre-Reacted Glass, PEG-400 DMA: Polyethylene Glycol 400 Dimethacrylate, BPA: Bisphenol A, MID: Minimally Invasive Dentistry, CHX: Chlorhexidine, WHO: World Health Organization, MIC: Minimum Inhibitory Concentration, SDF: Silver Diamine Fluoride
The most prevalent oral illness in the world is still dental caries [1]. Dental caries, often known as decay, is a condition that affects the hard tissues of the teeth and has a complex origin. Small surface roughness or subsurface demineralization is the first symptom, followed by cavitation, pulp involvement, swelling, abscess, and systemic symptoms [2]. In the history of conservative dentistry, amalgam has been a frequently utilized restorative material. However, because of the elevated risk of mercury leakage and amalgam phobia, the European Union outlawed the use of amalgam dental fillings in children under the age of 15 and a pregnant and lactating woman in 2017 after the Minamata convention was completed in 2013 [3]. As the demand for tooth-colored restorations increased and concerns about the potential toxicity of amalgam arose, dental resin composites were developed as alternatives to, or replacements for, silver amalgam. Since then, the field of composite dental restorations has continued to evolve [4].
Resin composites, the material of choice for direct restorations, have to be replaced more frequently by dentists because of their rising failure rates of the incidence of carious lesions existing at the margins of resin composite restorations, new dental restorative materials have been developed [5]. Current research has shifted toward the development of advanced restorative materials with aim of controlling the initiation of caries and promoting remineralization in its early stages, Depending on that, Bioactive materials are developed, they can effectively restore function because they are mechanically robust enough to support the masticatory and functional load [6]. They have the ability to release ions, especially those found in tooth minerals, that promote remineralization in the surrounding dental structure and avert the chemical imbalance that causes tooth mineral loss [5]. So these relatively new restorative bioactive materials fight the onset of secondary caries by combining the mechanical and aesthetic qualities of resinous materials with the antibacterial qualities of glass ionomers and the ability to remineralize tooth structure. This means that these bioactive materials not only restore form and function but also participate biologically by repairing diseased and damaged tissues leading to minimize the incidence of secondary dental caries [7]. Repairing decaying teeth using restorations that mimic living tissues (such as enamel, dentin, bone, cementum, etc.) in terms of appearance, function, and strength is known as bioactive materials [8]. These bioactive restorative materials is consistent with biologically oriented dental treatment approaches so they align the guidelines promoting minimally invasive techniques [9].
Different terminologies and classifications such as "biomimetic," "bioactive," "ion-releasing," and "smart "biomimetic," "bioactive," "ion-releasing," and "smart materials," have emerged based on the various interactions between the materials and the tooth substrate [10]. However, these terms remain a subject of ongoing debate, particularly among scientists, clinicians, and dental corporate members [11].
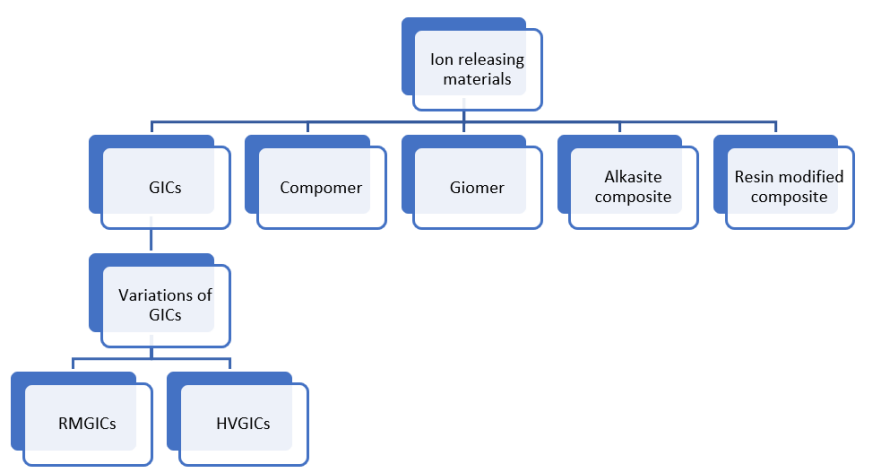
1. Glass Ionomer Cements (GICs)
As a remarkable innovation in 1969, Wilson and Kent created the dental material group known as glass ionomer cements (GICs) [12]. GICs first introduced as glass polyalkenoate cements consist of a degradable glass and a polyacrylic acid. Although the term GIC did not precisely describe its chemical composition, it had been adopted and accepted by most of dental clinicians. Conventional GIC was defined by McLean as ‘a cement that consists of basic glass and an acidic polymer, which sets by an acid–base reaction [13].
Glass ionomer restorative material has a number of important characteristics, including the capability of chemically adhere with tooth structures, acceptable color and translucency, along with the release of fluoride ions exhibiting an anti-cariogenic effect [12].
1.1 Chemistry of GICs
Fluoroaluminosilicate (FAS) glass powder and an aqueous solution of polyalkenoic acids -carboxylic acids-are the main ingredients of GICs. Polyacrylic acid makes up the majority of the aqueous fraction. To make handling easier, the solution could also include less viscous polyacids such maleic and itaconic acids. To increase the powder's radiopacity, other components like Barium (Ba) and Strontium (Sr) salts could be added, As well, Tartaric acid could be incorporated into the liquid component to improve handling qualities and extend working duration [12]. Conventional GICs, or those in their original classical form, are made from a powder-liquid mixture via an acid-base reaction. These are intricate substances that differ greatly in composition between formulations [14].The initial setting process is a gelation reaction between the constituents, and the cross-linking of the polymeric chains causes the composite to bind. The unreacted glass particles that serve as fillers in the silica (SiO2) gel matrix are then bound [12].
The acid-base reaction results in formation of silicic gel on the surface of the partially reacted FAS fillers, which helps to fix them firmly to the matrix and prevent hydrolysis by significantly increasing the cement's insolubility [14]. The cross-linking of the polymeric chains of the polyacid component (cross-linked acrylate matrix) with calcium and aluminum ions found in the powder component causes the resulting composite to harden. GICs set in two to three minutes, but the chemical process that results in full hardening can take place over the next twenty-four hours.
Typically, fluoride and sodium ions stay unreacted within the matrix and do not undergo any chemical reactions [12].
As Polyacrylic acid (PAA) is the primary ingredient in the liquid phase of traditional GICs, the materials' mechanical and physical characteristics are impacted by the composition and structure of PAA. For example, the conformation and flexibility of polyelectrolyte chains in polyacrylate salt matrix controlled the degree of polymer cross-linking and water movement across the cement during cement maturation. Water is also known to be necessary for the acid-base neutralization reaction that allows fluoride and other ions to be released from fluoro-alumino-silicate glass. The quantity of unreacted glass particles that serve as reinforcing fillers and increase the mechanical strength of GICs was similarly impacted by the degree of the acid-base reaction [15].
It has been suggested that the GIC's surface should be coated with petroleum jelly, light-cured bonding resins, CopalTM varnish, or a proprietary varnish. For conventional GICs, a novel resin covering has recently been created that is distinguished by the inclusion of nanofiller particles [16-18]. A highly distributed nanofilled resin coating called EquiaForte coat (GC Corp) was developed to enhance the GIC's wear resistance and marginal sealing [19].
1.2. Advantages and Drawbacks of GICs
GICs have demonstrated good secondary caries-inhibiting properties because of their antibacterial activity, low pH before setting, and long-term fluoride release. Furthermore, they can chemically adhere to the tooth structure by chelating calcium ions (Ca2+) in the apatite of enamel and dentine and the carboxyl group of acid polymeric chains and hence, the need for further adhesive systems was eliminated. Moreover, owning to the ion- releasing property, GICs may increase remineralization in adjacent proximal caries and reduce the chance of subsequent carious lesions progression [7]. Furthermore, GICs have been shown to exhibit antimicrobial effects as well as influence plaque formation [19]. In addition to the aforementioned characteristics of GICs, they also offer several other advantageous qualities including acceptable biocompatibility, low shrinkage, low marginal leakage, and a coefficient of thermal expansion comparable to that of natural tooth tissue. Despite these significant features, traditional versions had notable limitations such as poor color stability, moisture sensitivity, low fracture and abrasion resistance, and undesirable aesthetics. These drawbacks impair the material's physical characteristics limiting its application in regions subjected to high occlusal stresses [20]. Therefore, research has been conducted to further enhance its characteristics [12]. Moreover, the minimal degree of ion release that current GICs achieve may not be sufficient to stop tooth structure demineralization when exposed to acids from the biofilm on a regular basis. These factors have led to the creation of new materials or the alteration of existing ones in order to provide a more dependable anti-cariogenic action. The increased release of fluoride and/or other ions, such as calcium and phosphate, is a characteristic of these new materials that may aid in remineralization or stop demineralization following acid exposure [19].
1.3. GIC variations
1.3.1 Resin Modified Glass Ionomers (RMGIs): To address some of the limitations of GICs, a resin system was incorporated into the material to create a hybrid that combines the benefits of resin with the properties of GICs. This innovation was first described in a patent application in 1988, and subsequently introduced by Mitra in 1989 as a novel advancement [13]. RMGIs have the same fundamental composition as GICs but include an additional monomer component known as 2-hydroxyethyl methacrylate (HEMA), which acts as a ‘co-solvent’ to maintain all constituents in one phase, along with an initiator system (camphorquinone, CQ). These materials undergo setting by both an acid-base reaction and addition polymerization, resulting in a complex structure that is the outcome of two concurrent yet harmoniously balanced processes. However, it is essential to strictly adhere to the manufacturer’s instructions regarding irradiation duration to ensure the reliability of the final set material [21]. Nonetheless, certain disadvantages have been identified, notably plasticization and mechanical weakness, primarily due to the high water uptake associated with the hydrophilic nature of the incorporated HEMA monomer. Increased water sorption renders the polymer matrix more flexible, compromising the material’s physical proprieties [13]. They are therefore utilized as bases and liners in Class V restorations, as well as in Class I, II, and III restorations, all of which are mostly in the primary dentition. Additional applications include bonding agents for orthodontic brackets and fissure sealants [21].
1.3.2 High Viscous Glass Ionomers (HVGIs) and Hybrid Glass (ex Equiaforte): Later, conventional GICs were strengthened by altering the powder/liquid ratio, particle size, and distribution introducing highly viscous glass ionomer cements (HVGIC). HVGICs were launched to the dental market with the purpose of decreasing the moisture sensitivity of GIC in the early stages of hardening, increasing their hardness and abrasion resistance allowing them to be utilized in areas subjected to high stress-bearing forces [20,22]. These recently-introduced HVGICs have the same hardening mechanisms as traditional GIC. Besides, they possess superior abrasion resistance, fracture toughness, flexural strength, and moisture sensitivity as compared to traditional GIC. Furthermore, having a faster setting reaction prevents early water contact to exert any adverse effects on their physical characteristics [20].
A novel HVGIC restorative system (EQUIA; GC Europe, Tokyo, Japan) was introduced in 2007 [20]. It was optimized by increasing the molecular weight of the polyacrylic acid along with the incorporation of ultrafine, uniformly distributed highly reactive glass particles giving it enhanced mechanical features [19]. It was introduced to be used as a permanent substitute for resin composites in restoring Class I, II, and V cavities. By merging both benefits of HVGIC and a surface coating resin, better surface characteristics are obtained, gaps created by the material and finishing processes and surface irregularities are filled to create a smooth surface, moisture sensitivity is decreased during the early stages of hardening, resistance to fracture and abrasion is increased, and mechanical properties are improved [20]. the manufacturer asserts that it could be utilized in load-bearing posterior proximal restorations [19].
Other Bioactive Materials
2. Compomer
The words "composite" and "ionomer" are combined to form the term "compomer." These materials are sometimes known as polyacid-modified composite resins, in addition to this general term. These composites differ significantly from resin composites in two ways: one uses inorganic fillers, while the other uses organic monomers [14]. Nowadays, Compomers (e.g., Dyract®) are the first choice for restoring primary teeth. In contrast, composites should be used as a long-term filling material in permanent dentition because of their higher wear resistance and compressive, flexural, and tensile strength [23]. Compomers share approximately the same organic monomeric components of conventional composite including (bis-GMA) triethylene glycol dimethacrylate (TEGDMA) and (UDMA). Most of these formulations contain photoinitiators like camphorquinone because they are photopolymerizable. What distinguishes Compomer is the incorporation of a small proportion of functional monomers with carboxylic acid (–COOH) groups to the chemical composition, which is why they are also referred to as ‘polyacid-modified composite resins’ [14]. Due to that, the creation of compomers, or polyacrylic/polycarboxylic acid modified composites, is considered a combination of the benefits of composites (aesthetics) and glass ionomer cements(simple application) [23]. Regarding the inorganic filler components, The presence of silanated reactive FAS fillers in addition to silanated nonreactive quartz or silica fillers, which make up the majority of the matrix, is the second distinctive feature of compound. Under specific circumstances, these reactive fillers can bind to the resin matrix, improving its mechanical properties and releasing fluoride ions. These materials are available as reusable tubes or single-use compules. Photopolymerization is what starts these materials' setting reaction. Compomers initially exhibit mechanical characteristics that are somewhat similar to those of resin composites, but when the fillers hydrolyze and solubilize, their performance gradually deteriorates [14].
During a 6-month follow-up, one study examined the impact of conventional GIC versus compomer on Class I restorations. The result of this study showed that there was little to no difference in restoration failure between traditional GIC and compomer for Class I posterior restorations. On the other hand, according to the review of L. Pilcher et al, compomer might make restoration failure more likely [24]. Compomers are advised for primary carious molar restorations that are occlusal (Class I) or occluso-proximal (Class II) [25].
3. Giomer
Glass ionomers and composite resins have been merged by manufacturers. Based on pre-reacted glass (PRG) technology, Giomers are created, in which fluorine-containing glass particles and polymer-containing acid undergo an acid–base reaction in the presence of water to form a glass-ionomer phase prior to dispersal into the resin [26].
Pre-reacted glass fillers could be categorized into two types: full reaction type PRG (F-PRG) fillers and surface reaction type PRG (S-PRG) fillers. In F-PRG fillers, the surface as well as the core of the filler particle reacts completely with polyacrylic acid and hence, a large amount of fluoride is released. While In S-PRG fillers, only the surface of the filler reacts with polyacrylic acid, leaving the glass core intact [27,28]. When compared to traditional and resin-modified glass ionomers, giomers provide better clinical handling and physical attributes while maintaining the aesthetic qualities of resin composites [29]. In addition to that, it has been determined that the glass matrix plays a significant influence in fluoride release since studies have demonstrated that the rate of fluoride release in Giomer is higher than that of composite and compomer [30]. As well, Yap and Mok [31] reported that Giomer had shown superior surface smoothness in comparison to GIC after finishing procedures. With more enhancements in the filler structure, the second generation of Giomer exhibits exceptional qualities particularly in replicating the optical characteristics of tooth structure [19]. As a result, Giomers have demonstrated efficacy in treating Class I, II, and V lesions throughout the short to long term [29].
4. Alkasite composite
A new era of restorative dentistry begins with "alkasite" restorative material. In essence, it is a composite resin subgroup. It is a brand-new direct posterior bulk-fill restorative substance. An alkaline filler that can release ions that neutralize acid is used in this new material [32].
Alkasite is packaged separately in liquid and powder form. The liquid contains dimethacrylates and initiators, whereas the powder contains additional initiators, colors, and alkaline glass fillers. These chemicals release calcium, hydroxide, and fluoride ions that neutralize acid when the pH of the oral cavity decreases [33].
The ability to discharge hydroxide, calcium, and fluoride ions from its alkaline (calcium fluoro-silicate glass) filler is an alkasite material's primary benefit. The presence of hydroxide ions on the material's surface may be crucial in neutralizing the acids that cariogenic bacteria create [34].
A combination of urethane dimethacrylates, including UDMA, DCP, an aromaticaliphatic-UDMA, and PEG-400 DMA, make up the monomer matrix of this novel bioactive substance [35]. The UDMA-based resin matrix gives a natural translucency, enabling it to merge beautifully with teeth [36]. It is a self-curing substance that can optionally be light-cured, increasing the restoration's strength and lifespan. Shear bond strength, microtensile bond strength, compressive strength, and fracture resistance were found to be comparable to composite resins and higher than glass ionomer-based materials in earlier investigations assessing this unique alkasite material [33,37]. Furthermore, it was discovered that the material's microleakage at the enamel–restoration junction was less than that of composite resins [33].
Based on the clinical trial by Al-Salamony et al. [36], both alkasite and RMGI showed good clinical performance in Class V carious lesions; alkasite restorations can effectively replace RMGI for cervical restorations; and alkasite may be a better option in particular clinical scenarios, such as for high-risk patients who need wear resistance and fluoride release [36].
Ion releasing Composite
ACTIVA™BioACTIVE Restorative™ (Pulpdent, USA) is marketed as the first bioactive dental material with an ionic resin matrix, a shock-absorbing resin component, and bioactive fillers that replicate the physical and chemical characteristics of natural teeth [38]. They are free of bisphenol A glycol dimethacrylate (Bis-GMA), bisphenol A (BPA), and its derivatives, and they comprise patented bioactive ionic and moisture-friendly resin, patented rubberized resin, and reactive glass fillers. Three chemical reactions the acid-base reaction, light polymerization, and chemical curing combine to form their setting process [39]. It was recently introduced to the market as a hybridization between a RMGI and a resin composite. A mechanism by which the material reacts to pH cycles and actively participates in the release and recharge of calcium, phosphate, and fluoride ions underlies the bioactive qualities of ACTIVA™ products. They are thought to chemically attach to the tooth structure, sealing the cavity borders and reducing the creation of gaps at the tooth-restoration interface and microleakage [38].
Activa BioActive composite was selected as the experimental comparator to SDR Bulk-fill (manufacturer to be added) due to its high depth of light cure (4 mm) and low polymerization shrinkage (1.7%), which facilitates the application of larger increments, thereby reducing clinical application time [40]. Moreover, this product contains fluoride and silica ions, which may act as unpolymerized monomers following the polymerization process [39].
Due to these characteristics, ACTIVA is considered well-suited for restoring Class I / II cavities in primary molars. Activa BioActive composite exhibits high mechanical resistance and bioactive properties that make it easier to use in clinical settings than traditional composites and glass ionomer cements [41,42]. After a year of follow up, the Activa BioActive composite performed as the bulk-fill composite [40]. Additionally, according to the manufacturers’ claim, the fluoride-releasing capability of this material makes it particularly suitable for patients with high caries risk and in clinical scenarios where proper isolation cannot be achieved [40].
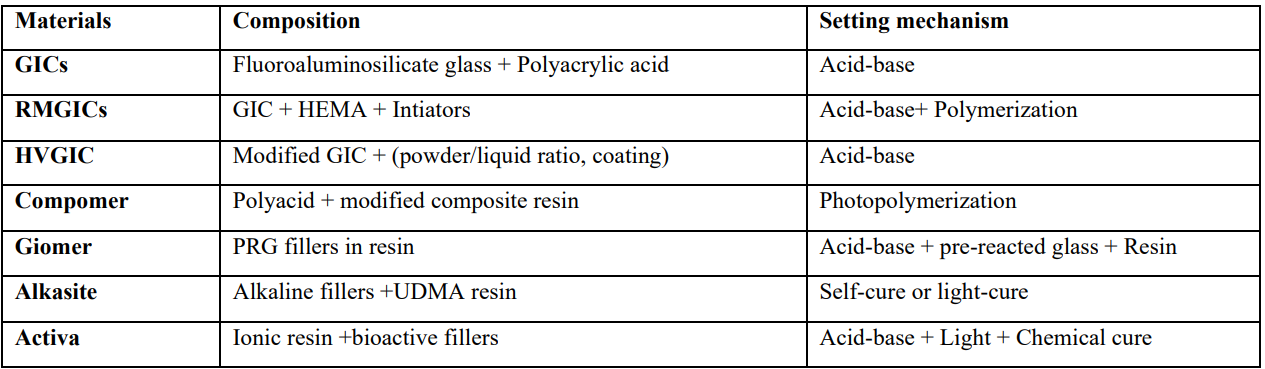
Table 1
Bioactive Properties Role on Material Development
Beyond the essential role of materials science in laying the foundation of bioactive dental materials, researchers today are increasingly adopting interdisciplinary techniques to expand the scope of biomaterials and enhance their clinical applications [11].
According to Mocquot et al.'s critical examination, [43] the definition of bioactivity depends on its clinical application and can include everything from the capacity to generate apatite precipitation on the surface of enamel and dentine to cellular effects that are sustained by the release of bioactive molecules and ions [43].
This term is consistent with the statement of the 2022 FDI World Dental Federation Policy; defining bioactive dental restorative material is one that can work chemically, biologically, or by combining both [9].
How Caries Process Occurs
Dental caries is a multifactorial disease primarily caused by three key elements: oral bacteria within dental plaque, the presence of fermentable carbohydrates, and an exposed tooth surface. Additionally, several contributing factors such as dietary habits, the quality and quantity of saliva, tooth morphology, surface characteristics, and oral hygiene practices can influence the rate of tooth decay, either accelerating or slowing it [2]. Dental caries is best described as a dynamic process that arises when an imbalance in the equilibrium between tooth minerals and the dental biofilm occurs [44]. More specifically, the carious lesion results from metabolic process within dental plaque which is formed of a sticky biofilm that contains various bacterial species. When the tooth surface is covered by dental plaque, the embedded acidogenic bacteria - mainly Streptococcus mutans In such an acidic environment, the mineral component of the tooth, predominantly hydroxyapatite, begins to dissolve, initiating the demineralization process. In a healthy oral environment, the buffering capacity of saliva, along with its calcium and phosphate ion content, helps to neutralize this acid and reverse demineralization through a process known as remineralization. This delicate balance ensures the continued deposition of hydroxyapatite, maintaining tooth integrity by replacing minerals lost through dissolution [46]. However, when the conditions favor demineralization over remineralization, mineral loss from enamel becomes visible as a white spot lesion. If left untreated, the lesion progresses, leading to a loss of enamel continuity in the form of a cavity that can eventually extend into the dentine [44].
How to Control Bacterial Progression
Early detection, precise diagnosis, halting the progression of the disease, and limited restorative intervention are essential for the effective management of dental caries. A contemporary kind of dental care called minimally invasive dentistry (MID) places a strong emphasis on maintaining healthy tooth structure while successfully treating dental cavities. Achieving long-lasting restorations with little intervention and a low risk of secondary caries development is the aim of MID [1].
Another crucial element of MID is the application of adhesive restorative materials, like glass ionomer cements. These substances can lessen the chance of microleakage and secondary caries while strengthening the link with the tooth structure [47-49].
The type of restorative material may arrest caries progression. Compared to glass ionomer cements and amalgam, dental plaque accumulated significantly higher in resin-based restoration materials. Therefore, after therapy, pulp irritation and recurrent and secondary caries may be caused by residual bacteria or invasive bacteria along the tooth/restoration contact [49,50]
After selective caries excavation, it is advised to clean the cavity with an antimicrobial solution to control any live bacteria that may still be present in the dentinal tubules [51]. Chlorhexidine (CHX) is thought to be the most popular cavity cleaner [52,53]. A broad-spectrum antibacterial, CHX inhibits both Gram-positive and Gram-negative bacteria, hence preventing the growth of any remaining germs [54]. According to earlier research, CHX strengthens resin composite bonds, protects the collagen matrix, and stops degradation at the resin–dentin interface [51,55].
To lessen cariogenic bacterial colonization on the tooth/restoration interface and the growth of leftover bacteria in the cavity after preparation, the restorative system should have an antimicrobial effect [1]. Incorporating antimicrobial agents or antibacterial monomers into the dental material formulation, altering the material composition to incorporate organic nanoparticles that promote remineralization and decrease demineralization, or employing inorganic nanoparticles like: calcium, fluoride, hydroxyapatite, bioactive glass, silver, zinc, magnesium, and copper oxides that actively inhibit the activity of oral microorganisms are some ways to accomplish an anticariogenic activity [56,57]. To achieve that, A new class of bioactive and therapeutic materials is being developed.
Factors Affecting Anticariogenicity
Diet control, pH of oral environment, good dental hygiene, antibacterial treatments and fluoride intake have long been the cornerstones of caries prevention, anticariogenicity and health promotion.
1. Diet control
Conveying the importance of oral hygiene for preserving quality of life remains a persistent challenge for oral health professionals. Complementing these strategies, dietary guidance plays a key role, with emphasis on reducing the intake of free sugars and fruit juices, in line with the most recent World Health Organization (WHO) recommendations for adults and children [58].
The consumption of free sugars should not surpass 10% of daily calorie intake, or less than 50 grams. Compared to those who consume more sugar, Individuals who consume less sugar have lower levels of caries-related species in their saliva and supragingival plaque, which increases anticariogenicity [59].
2. pH of oral environment
In order to support health-associated communities of beneficial oral bacteria and enhance the anticariogenicity, future preventative technologies should minimize the frequency and severity of low pH episodes in the tooth biofilm and maintain the pH near neutrality [58]. A pH of 5.5, known as the critical pH, is necessary for enamel breakdown to take place. Furthermore, the concentration of acids, their amount, their mineral chelating qualities, exposure duration, and temperature all have a significant role in the disintegration of enamel. Another important factor in the disintegration of enamel is saliva, including its composition, flow rate, and buffering ability [60]. The so-called critical pH and acid dissolution of the tooth have received a lot of attention [61]. Almost at the expense of all other possibilities, researchers have spent decades studying neutralizing acid as a way to stop the growth of carious lesions [62].
Such a method seems to ignore the fact that saliva has more than enough buffering power to neutralize any acid produced in the oral cavity. Why, then, does neutralization not take place? What causes the alkaline mouth to develop lesions? Simply put, the acid created in the depths of plaque that has accumulated on tooth surfaces is inaccessible to saliva. Naturally, plaque is a biofilm, and by definition, all biofilms have a matrix [63]. Bicarbonate and other potentially therapeutic substances cannot diffuse into plaque due to the matrix's chemical and physical characteristics [64]. Few organisms in dental plaque can convert sucrose into insoluble glucan, despite the fact that many of them can quickly produce acid. Streptococcus mutans is the best example of a plaque initiator and a strong acid generator. Therefore, it would seem that a careful reexamination of S. mutans' involvement in the etiology and pathophysiology of dental caries is necessary [62].
3. Oral hygiene
Caries and oral pathogens are effectively reduced by maintaining good dental health and good oral hygiene, consequently strengthening the anticariogenicity [65].
4. Antibacterial treatment and bioactive materials
Nowadays, a medicinal approach has replaced the traditional surgical approach to caries control. The use of Chlorhexidine cavity cleaner, fissure sealants and bioactive materials such as antibacterial agents, fluoride therapy, calcium phosphate products, peptides, and bioactive glass are all common components of this strategy. The materials' bioactive components have beneficial effects on the mouth environment and encourage both caries control and anticariogenicity [5]. In a simulated chewing model, Garcia et al. [66] sought to assess the wear behavior and surface quality of dental bioactive resins and compare them to a resin devoid of bioactive ingredients. Through the release of ions like calcium, phosphate, fluoride, zinc, magnesium, and strontium, bioactive materials might lessen caries lesions on the marginally sealed teeth. Such ions have an impact on the nucleation, epitaxial development, and dissolution balance of hydroxyapatite crystals [66].
5. Fluoride intake
As mentioned before; Fluoride enhance the remineralization, reduce demineralization and preventing caries process. So its presence enhances anticariogenicity [67]. High-fluoride varnishes applied professionally are useful for preventing both primary and secondary cavities. using fluoride toothpaste every day is the cornerstone of basic caries prevention for all age groups [58]. While prospective cohort studies provide support for regular fluoride exposure starting with the eruption of the first tooth builds on solid data from randomized controlled trials [58]
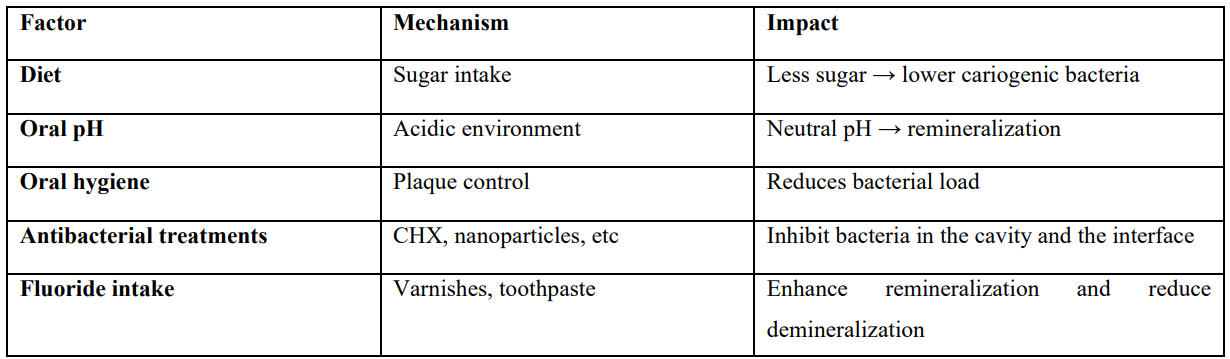
Table 2
Evidence on the Bioactive Materials Effects on the Oral Biofilm and the Anticariogenic Effect
1. Conventional glass ionomer
Dimkov et al [68]. investigated in their study the antibacterial activity of conventional glass ionomer cement against three distinct strains of microorganisms both alone and after adding 1, 2, and 3% benzoalkonium chloride and cetylpyridinium chloride. The study reported that, without the addition of antimicrobial substances, the glass-ionomer cement either creates very tiny inhibitory zones or none at all.
2. Resin modified GI
Saputra et al [69]. sought in this study to observe the growth inhibition zone of Streptococcus mutans in order to ascertain the antibacterial qualities of propolis, a natural product, in a mixture of resin-modified glass ionomer cement. The findings of this study revealed that (HEMA), which was included in the resin-modified glass ionomer liquid component, may have helped to promote the low pH and contributed to the antibacterial qualities. Therefore, resin-modified glass ionomer has a distinct inhibitory zone and antibacterial activity of its own, thereby playing a part in preventing secondary caries.
3. Highly viscous glass ionomer
Bathla et al [70]. intended in this in vitro investigation to evaluate four distinct GIC cements' antibacterial capabilities against S. mutans and L. acidophilus. One of these glass ionomer modifications was HVGIs. The result was that HVGIs was one of two materials that exhibited the strongest antibacterial activity for 48 hours which indicates maximum degree of secondary caries prevention.
Brkanović et al [71]. determined the wear rate of HVGICs (Equia Forte HT Fi) with or without coating and comparing it to another HVGICs (Fuji IX GP) under acid load or pH neutrality conditions. Results reported that coating Equia Forte with the Equia Forte coat raised the resistance of Equia Forte restoration to acid load reflecting that its antibacterial effect would be raised respectively along with its ability to prevent secondary caries.
4. Giomer
Feiz et al [30]. investigated the antibacterial activity of a number of fluoride-containing restorative materials one of them was giomer. Up to day 21, the minimum was associated with Giomer (Beautifil), which was constant. So Giomer has minimum degree of antibacterial effect and consequently secondary caries prevention.
5. Alkasites composite
Mohamed et al [72]. compared the antibacterial properties, microleakage, and microtensile bond strength (μTBS) of bulk fill resin composite with alkaline fillers (alkasite), such as ytterbium trifluoride and barium aluminum silicate glass, to incremental nanohybrid resin composite. When compared to chlorhexidine, resin composites containing alkaline fillers showed antibacterial activity yet it was modest.
6. Ion releasing Composite
Bagchi et al [73]. evaluated antibacterial qualities of ACTIVA BioACTIVE, Beautifil Flow Plus X, and Filtek Z250 XT in the current in vitro study. The conclusion was that ACTIVA BioACTIVE restorative material exhibited the best level of antibacterial effectiveness against S. mutans and the best level of secondary caries prevention.
Chaudhari et al [74]. compared the bactericidal activity of bioactive restorative composites (one of them was Activa BioACTIVE ) in vitro. The study concluded that Activa BioACTIVE restorative material had the highest antibacterial activity against S. mutans.
Methods of Evaluate Antibacterial Effect
1. Agar diffusion based screening of antimicrobial activity
The history of the agar diffusion technique begins in the late 18th century, when Beijerinck [75,76] used it for the first time. Since then, the approach has been modified in a number of ways to improve accuracy, such as by introducing the ditch plate technique and utilizing filter paper discs and wells (wells are made in the agar using a sterile cork borer or pipette tip). Additionally, variants like the agar plug (agar plugs impregnated with the test substance on the agar surface) and agar spot assays (a sterile inoculating loop is used to directly place a tiny volume of the test chemical onto the agar surface) have been created to give researchers more choices and flexibility [77].
The test material is positioned in the center of the thin layer of agar, to enable indirect contact measurement of inhibition zones [78]. These techniques rely on the diffusion of anti- microbial chemicals from tested sample into the surrounding agar medium by preventing the growth of the microorganism seeded on the agar surface. Researchers can determine the relative antibacterial potency of the test drug by measuring the ensuing zone of inhibition, which shows the area where microbial growth is blocked or hindered by the agent [77]. The agar overlay test only permits a qualitative assessment of cytotoxicity [78]. Agar plates are inoculated with a standardized inoculum of the test Microorganism in this technique. The agar surface is then covered with filter paper discs of the test substance at the appropriate concentration, each about 6 mm in diameter. Under the right circumstances, the Petri dishes are incubated. The sizes of the inhibitory growth zones are then determined. Researcher should illustrate the growth media, temperature, incubation time, and inoculum size to follow the Clinical and Laboratory Standards Institute criteria [79].
Numerous benefits set the disk-diffusion assay apart from other techniques, including ease of use, affordability, the capacity to test a vast array of bacteria and antimicrobial drugs, and the data' interpretability [79,80].
Although there are many benefits to the agar diffusion assay, researchers should be mindful of some of its drawbacks, such as its qualitative character, variability in diffusion, limited applicability, reliance on certain circumstances, and subjectivity in interpretation [81,82].
2. Bioluminiscence assay
Using genetically modified bacteria that express a light-emitting enzyme, usually luciferase, is the basic idea behind the bioluminescence assay [83,84].
3. Time kill kinetics
The time kill kinetics experiment exposes the test microorganism to varying concentrations of the antimicrobial agent over a predetermined period of time in order to assess antimicrobial activity [85].
4. Agar dilution and broth dilution methods
The minimum inhibitory concentration (MIC) of antimicrobial drugs can be found using two very comparable methods: the broth dilution method and the agar dilution method. MIC of an antibiotic agent is the concentration at which a certain bacterium can no longer grow visibly [86].
5. Resazurin assay
A flexible colorimetric test for determining the metabolic activity of living cells is the resazurin assay. The test's basic idea is that living cells' metabolic activity reduces the blue non-fluorescent dye resazurin to the pink fluorescent dye resorufin [87-89].
6. Antimicrobial gradient diffusion test
By integrating the basic ideas of antimicrobial agent dilution and diffusion, the antimicrobial gradient diffusion test provides an alternate method for assessing antimicrobial activity. the antimicrobial gradient diffusion test provides an alternate method for assessing antimicrobial activity. It is especially made to measure the Minimum inhibitory concentration, in contrast to agar diffusion assays which are mostly used as screening techniques [90-92].
7. Flow cytometry
Using lasers to identify and quantify the physical and chemical properties of cells or particles in a fluid stream is the fundamental idea behind flow cytometry [93].
8. Co-culture assay
Like the cross-streak approach, the co-culture assay allows direct interaction between two microorganisms by growing them simultaneously in a common environment. To verify the test microbe's inhibitory activity, the growth of the indicator organism is then measured and contrasted with a control. This technique is frequently used to evaluate the antibacterial activity of probiotic lactic acid bacteria against infections [94].
9. Impedance measurement
An effective method for identifying changes in the electrical characteristics of microbial cells in response to interactions with antimicrobial drugs is the impedance-based assay [95].
10. Other emerging techniques
Although they are less frequently used for high-throughput screening and large-scale assessment of antimicrobial activity, newer approaches like mass spectrometry and time-lapse microscopy, in addition to the previously mentioned methods, show great promise for the characterization and analysis of antimicrobial compounds [77].
Fluoride and Caries Prevention
Fluoride has long been recognized for its ability to prevent demineralization [96]. Fluoride ions can shift the balance in favor of remineralization through two main mechanisms; by directly inhibiting the metabolism of microbial cells within the dental biofilm, and by accelerating the remineralization process. On a biological level, the biochemical effects of fluoride on microbial cells are complex; however, they primarily involve the intracellular formation of hydrofluoric acid (HF), which impairs bacterial physiology and reduces viability [97]. In addition, fluoride can suppress a variety of characteristics linked to dental caries, such as enolase and enzymes involved in the synthesis of biofilm matrix, which would directly slow down glycolysis and indirectly lower bacterial sugar transport systems. Thus, it causes a sudden shutdown of sugar metabolism in oral biofilms, followed by a decrease in saccharolytic organisms [58]. This inhibition disrupts carbohydrate metabolism and reduces the number of Streptococcus mutans within the oral biofilm, resulting in fewer bacteria capable of metabolizing carbohydrates into caries-promoting acids. Consequently, this diminishes acid production and helps inhibit the development of dental caries [46].
On another hand, the hydroxyapatite crystals on enamel surface can react with fluoride ions to form fluorapatite (FA) and fluor hydroxyapatite (FHA) structures, such reactions reduce enamel solubility, inhibit demineralization, and promote remineralization [98], since the formed crystals are more acid-resistant than the original enamel structure [99]. Moreover, fluoride also reduces the capacity of saliva to dissolve enamel minerals by forming strong hydrogen bonds with water molecules [46]. In addition, Silver diamine fluoride (SDF), in contrast to other fluoride-based caries preventive treatments, can prevent and arrest coronal and root dentine caries at the same time [5]. Overall, these combined effects contribute significantly to halting the progression of carious lesions [46].
Current Evidence on F Release of Tested Materials (studies and researches)
1. Conventional glass ionomer
Rolim et al. [100], assessed F release from GICs both before and during the aging process and after recharging with F-dentifrices. This study reported that conventional GICs emit more fluoride than resin-based materials. Additionally, F release was improved by conventional GICs in comparison to the high viscosity one.
Nicholsona et al. [46] studied clinical implications of fluoride exchange via glass-ionomer dental cements. Authors concluded that, when it was held in aqueous conditions, it has been observed that several additional ions are also released in addition to fluoride such as phosphate, silicate, sodium, and aluminum. Similar to fluoride, each species elutes more in acidic conditions than in neutralized ones.
2. Resin modified GI
Francois et al. [14] sought to investigate fluoride-releasing restorative materials and new restorative materials In order to better understand their composition, setting reactions, mechanical, self-adhesive, and potential bulk-fill properties as well as their ion release capabilities. This study concluded that RM-GICs have release and recharge mechanisms that are comparable to those of conventional GICs, with a release peak observed upon insertion. However, it seem to emit less fluoride when compared to resin-free GICs, conventional GICs, and HVGICs.
3. Highly viscous glass ionomer
Vidal et al. [101], when evaluated the fluoride release of coat-free HV-GICs, alkasite composite and resin modified composite in their study, found that coat-free EQUIA Forte HT had the highest fluoride release overall while alkasite composite showed the greatest increase over time. In this investigation, ACTIVA BioACTIVE-RESTORATIVE showed the least amount of fluoride release.
Brzović-Rajić et al. [102] assessed the fluoride release from Glass Ionomer with Nano Filled Coat and Varnish. The findings of this in vitro study showed that, in comparison to the samples coated with two distinct agents for surface coating, the amount of released fluoride was substantially reduced in the samples coated with nanofilled surface coating agent compared to the samples coated with varnish and uncoated samples. This highlighted the effect of using a coat.
4. Giomer
Feiz et al. [30] compared and assessed the fluoride ion release and antibacterial activity of giomer, zirconomer, alkasite composite, and resin-modified GI taking into account the crucial role that fluoride played in preventing dental cavities by lowering the quantity and activity of cariogenic bacteria and launching novel fluoride-releasing restorative materials that were tooth-colored. This study resulted in that among the other groups, giomer released the least quantity of fluoride and had the least bactericidal action.
5. Alkasites
El-Adl et al. [103] evaluated the inhibitory effect of three ion-releasing restorative materials, which were alkasite restorative material, Ion releasing flowable composite liner, and Self-adhesive hybrid composite, on secondary caries by comparing their short-term fluoride release. The conclusion was that, in direct contact with the restoration, the three ion-releasing restorative materials demonstrated fluoride release and prevented the formation of secondary caries. The alkasite restorative material produced more fluoride when the pH was low.
6. Ion releasing composite
Tiskaya et al. [104] aimed in this investigation to describe the ion release, pH variations, and apatite forming capacity of two potentially bioactive composites: Ion releasing composite and alkasite composite. This investigation reported that, despite that Ion releasing composite release fluoride ions, the quantity of fluoride released was lower than that released from conventional glass ionomer cements with no appreciable variations in the fluoride release of it after being submerged in different pH solutions.
Factors Affecting F Release
1. Oral hygiene is one of the most important factors affecting F release. Research indicates that fluoride's cariostatic effects are primarily localized rather than systemic [105,106]. When combined with proper oral hygiene, this effect could be even more pronounced [107].
2. In addition to oral hygiene, pH determines the release of fluoride ions. F release is stimulated in acidic atmosphere more than neutral ones. Par et al. [108] used bioactive glass to create experimental composites and studied their release of calcium, phosphate, and fluoride, as well as pH variations and the precipitation of apatite following immersion. This study confirmed that the properties of the immersion media, particularly the pH, had a significant impact on the ion release profiles of both commercial and experimental restorative materials. Under acidic conditions (pH = 4.0), calcium, phosphate, and fluoride ions were released in a material-dependent manner. In artificial saliva (pH = 6.4), there is negative release of ions.
3. The composition of fluoride, whether it is free or complex, is the third factor affecting fluoride release and absorption. In acidic environments, more fluoride is released as complexes rather than free F-ions. In addition to that, hydroxyapatite absorbs complexed fluoride more easily than un-complexed fluoride [46].
Methods for F Evaluation
Over the last four decades, there has been a notable advancement in F analysis [109]. Despite the development of new techniques, assessing F remains neither easier nor more economical, since most methods are expensive, technically challenging, and of limited applicability [110]. Because of the inherent limits of these early procedures, erroneous assumptions were made despite the fact that early F analysis techniques allowed for important discoveries. Part of the reason for these inaccurate conclusions was that existing methods could not assess ionic fluoride in contrast to total fluoride [111,112].
In the last century, fluoride (F) analysis has evolved from simple colorimetric analysis, which yielded crude results and suffered from interference from other ions present in the samples, to more complex methods of analysis such as mass spectrometry, gas chromatography, ion chromatography, electroanalysis (Ion-selective electrode) [113,114] and spectrophotometry [115].
1. Spectrophotometry
In this method, bound fluorine is converted to free fluoride ions using UV photolysis. The generated fluorides are then used to determine the fluoride content using spectrophotometry, which is based on the destruction of the colored complex of zirconium–xylenol orange [115]. By reacting with a red zirconium dye, fluoride ions produce a colorless complex that reduces the red color in proportion to the fluoride concentration [116]. To prepare the SPADNS reagent, dissolve 958 mg (0.958 g) of SPADNS (Sulfanilic acid azochromotrop, Sigma Aldrich, USA) in 500 ml of deionized water. The zirconyl-acid reagent is obtained by dissolving 133 mg (0.133 g) of zirconyl chloride octahydrate, ZrOCl2–8H2O (Sigma Aldrich, USA), in approximately 25 mL of deionized water. Next, 350 mL of concentrated HCl is added, and the mixture is diluted to 500 ml using deionized water. Equal amounts of SPADNS solution and zirconyl-acid reagent are combined to produce the acid zirconyl-SPADNS reagent [117].
Advantages:-
This approach is affordable Compared to chromatography, straightforward, and easy [118]. in addition, spectrophotometry provides accurate and reliable results when appropriately prepared and It is suitable for analyzing drinking water and wastewater samples [119]. This method also offers increased repeatability and sensitivity [120] and it can also detect very low concentrations of fluoride [118,120].
Limitations:-
It requires chemical reagents and cannot measure fluoride directly [121,122], in addition, The results may be affected by interfering ions such as phosphate or aluminum and This methos has a very high pH sensitivity [123,124]. The preparation of reagents and calibration curve takes time and effort [120,124].
2. Mass spectrometry
The process relies on the formation of gas-phase ions that can be separated by magnetic or electrical means based on their mass to charge ratio [125].
Advantages:-
A potent method for single-cell analysis without labeling is mass spectrometry. It is incredibly sensitive, selective, and capable of simultaneously detecting numerous chemicals, which enables it to identify the structures of molecules of interest [125].
Limitations:-
Mass spectrometry has a number of drawbacks, including invasiveness, complicated data, semi-quantitativeness, and ion suppression [126].
3. Gas chromatography
For Gas chromatography analysis to perform effectively, the analytes must be able to transition from the liquid to the gas phase without breaking down [127]. in case of analysis of fluoride, By silylating the fluoride ion with trimethylchlorosilane, it can be transformed into trimethylfluorosilane. Headspace sampling has been used to do GC analysis of the derivative, which successfully lessens matrix interference [128].
Advantages:-
With the potential to refine quantification if the elemental composition and structure are partially understood, GC offers excellent resolution and approximate quantitation of unknown chemicals [129].
Limitations:-
Because trimethylfluorosilane is in the gas phase at room temperature, it is difficult to preserve and pretreat, which will negatively impact the precision and reproducibility of the results. Furthermore, the GC system may sustain significant damage from the HCl generated by the hydrolysis of trimethylchlorosilane [128].
4. Ion chromatography
A two column ovens, a flat-bed pen recorder , and a viable wavelength ultraviolet-visible detector were all part of the ion chromatography system. Only chemicals of analytical reagent grade were used, and dolomitized, distilled water was further filtered. Cetyldlmethyl-n-butylammonmm bronude, which was produced from cetyldunethylamme and n-butyl bromide, was synthesized using purified vvlth srlca gel. ODS rimmed columns were utilized to create dynamically coated columns [109,130].
Advantages:-
Short analysis times, small-volume sample analysis, high sensitivity and selectivity, simultaneous separation and detection of distinct ions or ions of the same element at different oxidation states are among of its primary benefits [131].
Limitations:-
Ion chromatography is a complex technique, Inappropriate processing techniques can negatively affect the IC system and produce less-than-ideal analytical results [131]. In addition, The fluoride ion's retention on the anion-exchange column is quite weak in ion chromatography, and organic acids can readily obstruct its detection [128].
5. Ion-selective electrode
A fluoride ion-selective electrode was utilized to determine fluoride ion activity utilizing the direct potentiometry method. The reference electrode was a standard silver chloride electrode. The electromotive force of a galvanic cell was measured using a typical laboratory ion meter. Every day prior to fluoride tests, the electrode was calibrated [132]. The measured potential linked to fluoride ion activity in solution is described by the Nernst equation [109].
Fluoride in other forms, such as when mixed to form a complex ion, cannot be detected. Before measuring fluoride in the storage solutions, the vast majority of studies add total ionic solubility acid buffer (TISAB) solution to forcefully decomplex all of the fluoride present [46]. Cyclohexylenedinitrilotetraacetic acid (CDTA), an Al ion masking agent found in all TISABs, preferentially combines with Al and releases F from complexes with this ion (Schamschula et al., 1985; Tusˇl, 1970). Commonly, TISABs are utilized in their commercially accessible forms, which are II, III, and IV. TISAB IV raises the pH of samples and standards to about 8.5, while TISAB II and III raise them to between 5 and 5.5 [112]. Therefore; addition of (TISAB) throughout analysis offers several benefits, including maintaining a constant background ionic strength, de-complexing F ions, and controlling the solution's pH (5.0–5.5) [133].
Advantages:-
Ion-selective electrode provide a simple and rapid method for fluoride determination and it allows accurate and reproducible measurements with low standard deviations [130,134]. ISEs can directly measure free ion concentrations without complex sample preparation [135]. In addition, it is affordable and require relatively simple instrumentation compared with chromatographic or spectroscopic techniques [136]. It provides high selectivity because of The electrode's superior selectivity. Additionally, this approach has the benefit of detecting even minute amounts of fluoride ion concentrations with this sensor [137].
Limitations:-
The fluoride ion can be easily disturbed by coexisting ions in the solution when using the selective-ion electrode approach, and the electrode's potential drift can also affect the result. So the electrodes require frequent calibration to maintain accuracy [128,134]. They are less reliable at very low ion concentrations, where the signal may become meaningless [138,140]. Interference from other ions may affect the selectivity and lead to inaccurate results [132]. They can show potential drift over time, which reduces stability and accuracy of long-term measurements [140]. Solid-contact ISEs may suffer from water layer formation at the interface, which destabilizes the signal [140].
Limited Data on the Anticariogenic Effect and Fluoride Release of the Tested Materials Compared to GICs and the Importance of Further Research
Many studies have been conducted on GIC because of its characteristics, which include antibacterial activity, ion leachability, biocompatibility, and the ability to stick to metals, enamel, dentine, and bone. In addition, a new era of bioactive materials has also emerged as a result of numerous attempts to enhance the characteristics of conventional GIC [141].
The majority of research on creating a new class of medicinal, bioactive, and antimicrobial resins is conducted in vitro studies. Furthermore, there hasn't been much focus on how to maximize the microecologic regulating effects of antimicrobial materials [142]. Further research is necessary to examine not just the antibacterial impact but also the released ions (such as fluoride). There is little research on these new ion releasing materials' capacity to prevent secondary caries formation and their potential for fluoride release. Consequently, there is a growing demand for additional research [103].
Conclusion
Bioactive restorative materials represent a significant advancement in modern dentistry by integrating mechanical, aesthetic, and biological functions. Materials such as HVGICs, compomers, giomers, alkasite composites, and bioactive composites like ACTIVA not only restore teeth but actively participate in remineralization, fluoride release, and inhibition of cariogenic bacteria. Despite promising in vitro and clinical evidence, further research is needed to quantify their long-term anticariogenic effectiveness, ion release dynamics, and ability to modulate oral biofilms in vivo. These materials align with minimally invasive dentistry principles and offer potential for more durable and biologically compatible restorative solutions.
References
- Abuljadayel R., Aljadani N., Almutairi H., Turkistani A. Effect of Antibacterial Agents on Dentin Bond Strength of Bioactive Restorative Materials. Polymers. 2023;15:2612. [PubMed.]
- Mathur VP., Dhillon JK. Dental Caries: A Disease Which Needs Attention. The Indian Journal of Pediatrics. 2018;85:202‒6. [PubMed.]
- Behlau A., Behlau I., Payer M., Leitinger G., Hanscho K., et al. Effects of Finishing on Surface Roughness of Four Different Glass‒Ionomer Cements and One Alkasite: In Vitro Investigation over Time Using Aging Simulation. Journal of Functional Biomaterials. 2024;15:325. [PubMed.]
- Worthington HV., Khangura S., Seal K., Mierzwinski‒Urban M., Veitz‒Keenan A., et al. Direct composite resin fillings versus amalgam fillings for permanent posterior teeth. Cochrane Database of Systematic Reviews. 2021;2021. [PubMed.]
- Melo MAS., Mokeem L., Sun J. Bioactive Restorative Dental Materials‒The New Frontier. Dent Clin North Am. 2022;66:551‒66. [PubMed.]
- Almulhim KS., Syed MR., Alqahtani N., Alamoudi M., Khan M., et al. Bioactive Inorganic Materials for Dental Applications: A Narrative Review. Materials. 2022;15:6864. [PubMed.]
- Conti G., Veneri F., Amadori F., Garzoni A., Majorana A., et al. Evaluation of Antibacterial Activity of a Bioactive Restorative Material Versus a Glass‒Ionomer Cement on Streptococcus Mutans: In‒Vitro Study. Dentistry Journal. 2023;11:149. [PubMed.]
- Singer L., Fouda A., Bourauel C. Biomimetic approaches and materials in restorative and regenerative dentistry: review article. BMC Oral Health. 2023;23. [PubMed.]
- Garoushi S., Peltola T., Siekkinen M., Hupa L., Vallittu PK., et al. Retention of strength and ion release of some restorative materials. Odontology. 2025;113:714‒23. [PubMed.]
- Zafar MS., Amin F., Fareed MA., Ghabbani H., Riaz S., et al. Biomimetic Aspects of Restorative Dentistry Biomaterials. Biomimetics. 2020;5:34. [PubMed.]
- Pires PM., Rosa TDC., Ribeiro‒Lages MB., Duarte ML., et al. Bioactive Restorative Materials Applied over Coronal Dentine—A Bibliometric and Critical Review. Bioengineering. 2023;10:731. [Ref.]
- Najeeb S., Khurshid Z., Zafar M., Khan A., Zohaib S., et al. Modifications in Glass Ionomer Cements: Nano‒Sized Fillers and Bioactive Nanoceramics. International Journal of Molecular Sciences. 2016;17:1134. [PubMed.]
- Agha A., Parker S., Patel MP. Development of experimental resin modified glass ionomer cements (RMGICs) with reduced water uptake and dimensional change. Dent Mater. 2016;32:713‒22. [PubMed.]
- Francois P., Fouquet V., Attal J‒P., Dursun E. Commercially Available Fluoride‒Releasing Restorative Materials: A Review and a Proposal for Classification. Materials. 2020;13:2313. [PubMed.]
- Panpisut P., Monmaturapoj N., Srion A., Toneluck A., Phantumvanit Prathip. Physical Properties of Glass Ionomer Cement Containing Pre‒Reacted Spherical Glass Fillers. Brazilian Dental Journal. 2020;31:445‒52. [PubMed.]
- McLean JW., Wilson AD. The clinical development of the glass‒ionomer cement. II. Some clinical applications. Aust Dent J. 1977;22:120‒7. [PubMed.]
- Nicholson JW., Czarnecka B. Kinetic studies of the effect of varnish on water loss by glass‒ionomer cements. Dent Mater. 2007;23:1549‒52. [PubMed.]
- Valera VC., Navarro MF., Taga EM., Pascotto RC. Effect of nail varnishes and petroleum jelly combinations on glass ionomer dye uptake. Am J Dent. 1997;10:251‒3. [Ref.]
-
Sobh E., Hamama H., Palamara J., et al. Effect of
CPP‐ACP modified‐GIC on prevention of demineralization in comparison to other fluoride‐containing restorative materials. Australian Dental Journal. 2022;67:220‒9. [PubMed.] - Balkaya H., Arslan S., Pala K. A randomized, prospective clinical study evaluating effectiveness of a bulk‒fill composite resin, a conventional composite resin and a reinforced glass ionomer in Class II cavities: one‒year results. Journal of Applied Oral Science. 2019;27. [PubMed.]
- Sidhu S., Nicholson J. A Review of Glass‒Ionomer Cements for Clinical Dentistry. Journal of Functional Biomaterials. 2016;7:16. [PubMed.]
- Friedl K., Hiller KA., Friedl KH. Clinical performance of a new glass ionomer based restoration system: a retrospective cohort study. Dent Mater. 2011;27:1031‒7. [PubMed.]
- Möhn M., Bulski JC., Krämer N., Rahman A., Schulz‒Weidner N. Management of Amelogenesis Imperfecta in Childhood: Two Case Reports. International Journal of Environmental Research and Public Health. 2021;18:7204. [PubMed.]
- Pilcher L., Pahlke S., Urquhart O., O’Brien KK., Dhar V., et al. Direct materials for restoring caries lesions. The Journal of the American Dental Association. 2023;154:e1‒e98. [PubMed.]
- Duggal M., Gizani S., Albadri S., Kramer N., Stratigaki E., Tong HJ., et al. Best clinical practice guidance for treating deep carious lesions in primary teeth: an EAPD policy document. European Archives of Paediatric Dentistry. 2022;23:659‒66. [PubMed.]
- Ozer F., Patel R., Yip J., et al. Five‐year clinical performance of two fluoride‐releasing giomer resin materials in occlusal restorations. Journal of Esthetic and Restorative Dentistry. 2022;34:1213‒20. [PubMed.]
- Neto CCL., das Neves AM., Arantes DC., et al. Evaluation of the clinical performance of GIOMERs and comparison with other conventional restorative materials in permanent teeth: a systematic review and meta‒analysis. Evid Based Dent. 2022. [PubMed.]
- Sunico MC., Shinkai K., Katoh Y. Two‒year clinical performance of occlusal and cervical giomer restorations. Oper Dent. 2005;30:282‒9. [PubMed.]
- Ozer F., Irmak O., Yakymiv O., Mohammed A., Pande R., et al. Three‒year Clinical Performance of Two Giomer Restorative Materials in Restorations. Operative Dentistry. 2021;46:E60‒E7. [PubMed.]
- Feiz A., Nicoo MA., Parastesh A., Jafari N., Sarfaraz D. Comparison of antibacterial activity and fluoride release in tooth‒colored restorative materials: Resin‒modified glass ionomer, zirconomer, giomer, and cention N. Dental Research Journal. 2022;19:104. [PubMed.]
- Yap AU., Mok BY. Surface finish of a new hybrid aesthetic restorative material. Oper Dent. 2002;27:161‒6. [PubMed.]
- Abdallah RM., Aref NS. Development of Newly Formulated Nanoalumina‒/Alkasite‒Based Restorative Material. International Journal of Dentistry. 2021;2021:1‒6. [PubMed.]
- Yazkan B., Celik E., Recen D. Effect of Aging on Surface Roughness and Color Stability of a Novel Alkasite in Comparison with Current Direct Restorative Materials. Operative Dentistry. 2021;46:E240‒E50. [PubMed.]
- Wiriyasatiankun P., Sakoolnamarka R., Thanyasrisung P. The impact of an alkasite restorative material on the pH of Streptococcus mutans biofilm and dentin remineralization: an in vitro study. BMC Oral Health. 2022;22. [PubMed.]
- Pai D., Anirudhmaadhava PA., Ginjupalli K. In Vitro Evaluation of Mechanical Properties of Cention N and Its Comparison with Resin Modified Glass Ionomer Cement (RMGIC) Restorative Material as Used in Primary Teeth. The Scientific World Journal. 2024;2024:1‒7. [PubMed.]
- Al‒Salamony H., Akah M., Naguib EA., Safwat OM. Clinical Evaluation of Bulk‒fill Alkasite Restoration vs Resin‒modified Glass Ionomer in Class V Carious Dent Lesions: 1‒year Randomized Clinical Trial. J Contemp Pract. 2024;25:1127‒34. [PubMed.]
- Yao C., Ahmed MH., Okazaki Y., Van Landuyt KL., Huang C Bart Van Meerbeek. Bonding Efficacy of a New Self‒Adhesive Restorative onto Flat Dentin vs Class‒I Cavity‒bottom Dentin. J Adhes Dent. 2020;22:65‒77. [PubMed.]
- Martínez‒Sabio L., Penate L., Arregui M., Veloso Duran A., Blanco JR., et al. Comparison of Shear Bond Strength and Microleakage between Activa™ Bioactive Restorative™ and Bulk‒Fill Composites—An In Vitro Study. Polymers. 2023;15:2840. [PubMed.]
- Vouzara T., Roussou K., Nikolaidis AK., Tolidis K., Koulaouzidou EA. Organic Eluates Derived from Intermediate Restorative Dental Materials. Molecules. 2020;25:1593. [PubMed.]
- Lardani L., Derchi G., Marchio V., Carli E. One‒Year Clinical Performance of Activa™ Bioactive‒Restorative Composite in Primary Molars. Children. 2022;9:433. [PubMed.]
- Frankl L., Hellman I. Symposium on child analysis. The ego's participation in the therapeutic alliance. Int J Psychoanal. 1962;43:333‒7. [PubMed.]
- Hickel R., Peschke A., Tyas M., Mjor I., Bayne S., et al. FDI World Dental Federation ‒ clinical criteria for the evaluation of direct and indirect restorations. Update and clinical examples. J Adhes Dent. 2010;12:259‒72. [PubMed.]
- Mocquot C., Attik N., Pradelle-Plasse N., Grosgogeat B., Colon P. Bioactivity assessment of bioactive glasses for dental applications: A critical review. Dent Mater. 2020;36:1116‒43. [PubMed.]
- Tulek A., Mulic A., Runningen M., Lillemo J., Utheim TP., et al. Genetic Aspects of Dental Erosive Wear and Dental Caries. International Journal of Dentistry. 2021;2021:1‒14. [PubMed.]
- Robati M., Yousefimanesh H., Shokuhi Far MR., Bagheri S. Effect of low‒level diode laser on streptococcus mutans and lactobacillus acidophilus growth: An invitro study. J Oral Biol Craniofac Res. 2022;12:396‒400. [PubMed.]
- Nicholson JW., Sidhu SK., 09-. Fluoride exchange by glass‒ionomer dental cements and its clinical effects: a review. Biomaterial Investigations in Dentistry. 2023;10. [PubMed] [PubMed.]
- Ericson D. The concept of minimally invasive dentistry. Dent Update. 2007;34:9‒10. [PubMed.]
- Frencken JE., Peters MC., Manton DJ., Leal SC., Gordan VV., et al. Minimal intervention dentistry for managing dental caries ‒ a review: report of a FDI task group. Int Dent J. 2012;62:223‒43. [PubMed.]
- Torres PJ., Phan HT., Bojorquez AK., Garcia‒Godoy F., Pinzon LM. Minimally Invasive Techniques Used for Caries Management in Dentistry. A Review. J Clin Pediatr Dent. 2021;45:224‒32. [PubMed.]
- Momoi Y., Hayashi M., Fujitani M., Fukushima M., Imazato S., et al. Clinical guidelines for treating caries in adults following a minimal intervention policy‒‒evidence and consensus based report. J Dent. 2012;40:95‒105. [PubMed.]
- Coelho A., Amaro I., Rascao B., Marcelino I., Anabela Paul., Jose Saraiva., et al. Effect of Cavity Disinfectants on Dentin Bond Strength and Clinical Success of Composite Restorations‒A Systematic Review of In Vitro, In Situ and Clinical Studies. Int J Mol Sci. 2020;22. [PubMed.]
- Elkassas DW., Fawzi EM., Ahmed El Zohairy. The effect of cavity disinfectants on the micro‒shear bond strength of dentin adhesives. Eur J Dent. 2014;8:184‒90. [PubMed.]
- Sheikh Hasani Y., Paryab M., Saffarpour A., Kharazifard MJ., Shahrabi M. The Effect of Disinfection with Chlorhexidine on the Shear Bond Strength of Equia Resin‒Modified Glass Ionomer Cement to Dentin in Permanent Teeth after Two Thermocycling Protocols. J Dent (Shiraz). 2017;18:265‒71. [PubMed.]
- Dursun E., Le Goff S., Ruse DN., Attal JP. Effect of chlorhexidine application on the long‒term shear bond strength to dentin of a resin‒modified glass ionomer. Oper Dent. 2013;38:275‒81. [PubMed.]
- Zheng P., Zaruba M., Attin T., Wiegand A. Effect of different matrix metalloproteinase inhibitors on microtensile bond strength of an etch‒and‒rinse and a self‒etching adhesive to dentin. Oper Dent. 2015;40:80‒6. [PubMed.]
- Chen L., Shen H., Suh BI. Antibacterial dental restorative materials: a state‒of‒the‒art review. Am J Dent. 2012;25:337‒46. [PubMed.]
- Sun Q., Zhang L., Bai R., Zhuang Z., Zhang Y., Yu T., et al. Recent Progress in Antimicrobial Strategies for Resin‒Based Restoratives. Polymers (Basel). 2021;13. [PubMed.]
- Pitts NB., Twetman S., Fisher J., Marsh PD. Understanding dental caries as a non‒communicable disease. British Dental Journal. 2021;231:749‒53. [PubMed.]
- Keller MK., Kressirer CA., Belstrom D., Twetman S., Tanner ACR. Oral microbial profiles of individuals with different levels of sugar intake. J Oral Microbiol. 2017;9:1355207. [PubMed.]
- Dash KC., Panda A., Bhuyan L., Ghosh B., Pal I. Dissolution of Enamel on Exposure to Various Commercial Beverages Available in India. The Journal of Contemporary Dental Practice. 2017;18:1009‒13. [PubMed.]
- Bowen WH. The Stephan Curve revisited. Odontology. 2013;101:2‒8. [PubMed.]
- Bowen WH. Dental caries – not just holes in teeth! A perspective. Molecular Oral Microbiology. 2016;31:228‒33. [PubMed.]
- Flemming H‒C., Wingender J. The biofilm matrix. Nature Reviews Microbiology. 2010;8:623‒33. [PubMed.]
- Bowen WH., Koo H. Biology of Streptococcus mutans‒derived glucosyltransferases: role in extracellular matrix formation of cariogenic biofilms. Caries Res. 2011;45:69‒86. [PubMed.]
- Stein C., Santos NML., Hilgert JB., Hugo FN. Effectiveness of oral health education on oral hygiene and dental caries in schoolchildren: Systematic review and meta‒analysis. Community Dent Oral Epidemiol. 2018;46:30‒7. [PubMed.]
- Garcia IM., Balhaddad AA., Ibrahim MS., Weir MD., Xu HHK., et al. Antibacterial response of oral microcosm biofilm to nano‒zinc oxide in adhesive resin. Dent Mater. 2021;37:e182‒e93. [PubMed.]
- Wassel MO., Allam GG. Anti‒Bacterial effect, fluoride release., and compressive strength of a glass ionomer containing silver and titanium nanoparticles. Indian J Dent Res. 2022;33:75‒9. [PubMed.]
- Dimkov A., Gjorgievska E., Nicholson JW., Kaftandzieva A. Antibacterial effects of conventional glass ionomer cement. Bratislava Medical Journal. 2016;116:31‒5. [PubMed.]
- Saputra SK., Sutantyo D., Farmasyanti CA., Alhasyimi AA. The effect of the addition of propolis to resin‒modified glass ionomer cement bracket adhesive materials on the growth inhibition zone of Streptococcus mutans. F1000Research. 2020;8:2105. [PubMed.]
- Bathla S., Dutta SK., Bagchi A., Priya CVP., Raj RKS., et al. Comparison of Antibacterial Activity of Various Additives to Glass Ionomer Restoration: An In Vitro Study. J Pharm Bioallied Sci. 2024;16:S189‒s91. [PubMed.]
- Brkanovic S., Ivanisevic A., Miletic I., Mezdic D., Jukic Krmek S. Effect of Nano‒Filled Protective Coating and Different pH Enviroment on Wear Resistance of New Glass Hybrid Restorative Material. Materials. 2021;14:755. [PubMed.]
- Mohamed NI., Safy RK., Elezz AFA. Microtensile Bond Strength., Marginal Leakage., and Antibacterial Effect of Bulk Fill Resin Composite with Alkaline Fillers versus Incremental Nanohybrid Composite Resin. European Journal of Dentistry. 2021;15:425‒32. [PubMed.]
- Bagchi A., Dolker T., Talukdar A., Sowmya K., Khan MM., et al. An in vitro Study on the Antibacterial Efficiency of the Various Composites in the Children: An Original Research. J Pharm Bioallied Sci. 2023;15:S192‒s5. [PubMed.]
- Chaudhari PR., Shashikiran ND., Gugawad S., Gaonkar N., Taur S., et al. Comparative evaluation of antibacterial efficacy different bioactive smart composites: An in vitro study. J Indian Soc Pedod Prev Dent. 2021;39:388‒91. [PubMed.]
- Smith KP., Richmond DL., Brennan‒Krohn T., Elliott HL., Kirby JE. Development of MAST: A Microscopy‒Based Antimicrobial Susceptibility Testing Platform. SLAS Technol. 2017;22:662‒74. [PubMed.]
- Wheat PF. History and development of antimicrobial susceptibility testing methodology. J Antimicrob Chemother. 2001;48 Suppl 1:1‒4. [PubMed.]
- Hossain TJ. Methods for screening and evaluation of antimicrobial activity: A review of protocols, advantages, and limitations. European Journal of Microbiology and Immunology. 2024;14:97‒115. [PubMed.]
- Pusnik M., Imeri M., Deppierraz G., Bruinink A., Zinn M. The agar diffusion scratch assay ‒ A novel method to assess the bioactive and cytotoxic potential of new materials and compounds. Scientific Reports. 2016;6:20854. [PubMed.]
- Balouiri M., Sadiki M., Ibnsouda SK. Methods for in vitro evaluating antimicrobial activity: A review. J Pharm Anal. 2016;6:71‒9. [PubMed.]
- Kreger BE., Craven DE., McCabe WR. Gram‒negative bacteremia. IV. Re‒evaluation of clinical features and treatment in 612 patients. Am J Med. 1980;68:344‒55. [PubMed.]
- Bonev B., Hooper J., Parisot J. Principles of assessing bacterial susceptibility to antibiotics using the agar diffusion method. J Antimicrob Chemother. 2008;61:1295‒301. [PubMed.]
- Liu Y., Li J., Du J., Hu M., Bai H., et al. Accurate assessment of antibiotic susceptibility and screening resistant strains of a bacterial population by linear gradient plate. Sci China Life Sci. 2011;54:953‒60. [PubMed.]
- Czieborowski M., Kemperman AJB., Rolevink E., Blom J., Visser T., et al. A two‒step bioluminescence assay for optimizing antibacterial coating of hollow‒fiber membranes with polydopamine in an integrative approach. Journal of Microbiological Methods. 2022;196:106452. [PubMed.]
- Robinson GM., Tonks KM., Thorn RM., Reynolds DM. Application of bacterial bioluminescence to assess the efficacy of fast‒acting biocides. Antimicrob Agents Chemother. 2011;55:5214‒20. [PubMed.]
- Xedzro C., Tano‒Debrah K., Nakano H. Antibacterial efficacies and time‒kill kinetics of indigenous Ghanaian spice extracts against Listeria monocytogenes and some other food‒borne pathogenic bacteria. Microbiol Res. 2022;258:126980. [PubMed.]
- Schumacher A., Vranken T., Malhotra A., Arts JJC., Habibovic P. In vitro antimicrobial susceptibility testing methods: agar dilution to 3D tissue‒engineered models. European Journal of Clinical Microbiology & Infectious Diseases. 2018;37:187‒208. [PubMed.]
- Chakansin C., Yostaworakul J., Warin C., Kulthong K., Boonrungsiman S. Resazurin rapid screening for antibacterial activities of organic and inorganic nanoparticles: Potential., limitations and precautions. Anal Biochem. 2022;637:114449. [PubMed.]
- O'Neill TE., Li H., Colquhoun CD., Johnson JA., Webster D., et al. Optimisation of the microplate resazurin assay for screening and bioassay‒guided fractionation of phytochemical extracts against Mycobacterium tuberculosis. Phytochem Anal. 2014;25:461‒7. [PubMed.]
- Travnickova E., Mikula P., Oprsal J., Bohacova M., Kubac L., et al. Resazurin assay for assessment of antimicrobial properties of electrospun nanofiber filtration membranes. AMB Express. 2019;9. [PubMed.]
- Berghaus LJ., Giguere S., Guldbech K., Warner E., Ugorji U., et al. Comparison of Etest, disk diffusion, and broth macrodilution for in vitro susceptibility testing of Rhodococcus equi. J Clin Microbiol. 2015;53:314‒8. [PubMed.]
- Cimen C., Siemer P., Sattler J., Voss A., Berends MS. Performance of disk diffusion, gradient test, and VITEK 2 for carbapenem susceptibility testing in OXA‒48‒like carbapenemase‒producing Enterobacterales: a comparative study. Journal of Clinical Microbiology. 2025;63. [PubMed.]
- Goldstein FW., Ly A., Kitzis MD. Comparison of Etest with agar dilution for testing the susceptibility of Pseudomonas aeruginosa and other multidrug‒resistant bacteria to colistin. J Antimicrob Chemother. 2007;59:1039‒40. [PubMed.]
- Lapinska B., Konieczka M., Zarzycka B., Sokolowski K., et al. Flow Cytometry Analysis of Antibacterial Effects of Universal Dentin Bonding Agents on Streptococcus mutans. Molecules. 2019;24:532. [PubMed.]
- Lodhi AF., Zhang Y., Adil M., Deng Y. Antibiotic discovery: combining isolation chip (iChip) technology and co‒culture technique. Applied Microbiology and Biotechnology. 2018;102:7333‒41. [PubMed.]
- Spencer DC., Paton TF., Mulroney KT., Inglis TJJ., et al. A fast impedance‒based antimicrobial susceptibility test. Nature Communications. 2020;11. [PubMed.]
- Ghilotti J., Fernández I., Sanz JL., Melo M., Llena C. Remineralization Potential of Three Restorative Glass Ionomer Cements: An In Vitro Study. Journal of Clinical Medicine. 2023;12:2434. [Ref.]
- Liao Y., Brandt BW., Li J., Crielaard W., Van Loveren C., et al. Fluoride resistance inStreptococcus mutans: a mini review. Journal of Oral Microbiology. 2017;9:1344509. [PubMed.]
- Zhang Q., Guan L., Guo J., Chuan A., Tong J., et al. Application of fluoride disturbs plaque microecology and promotes remineralization of enamel initial caries. Journal of Oral Microbiology. 2022;14. [PubMed.]
- Stifler CA., Wittig NK., Sassi M., et al. X‒ray Linear Dichroism in Apatite. Journal of the American Chemical Society. 2018;140:11698‒704. [PubMed.]
- Rolim FG., De Araújo Lima AD., Lima Campos IC., De Sousa Ferreira R., Da Cunha Oliveira‒Junior C., et al. Fluoride Release of Fresh and Aged Glass Ionomer Cements after Recharging with High‒Fluoride Dentifrice. International Journal of Dentistry. 2019;2019:1‒6. [PubMed.]
- Banic Vidal LS., Vecek NN., et al. Short‒Term Fluoride Release from Ion‒ Releasing Dental Materials. Acta Stomatol Croat. 2023;57:229‒37. [PubMed.]
- Brzovic‒Rajic V., Miletic I., Gurgan S., Peros K., Verzak Z., Ivanisevic‒Malcic A. Fluoride Release from Glass Ionomer with Nano Filled Coat and Varnish. Acta Stomatol Croat. 2018;52:307‒13. [PubMed.]
- El‒Adl ET., Ebaya MM., Habib E‒SE., Zaghloul NM. Comparative measurement of short‒term fluoride release and inhibition of caries around restoration by ion releasing restorative materials: an in vitro study. Scientific Reports. 2025;15. [PubMed.]
- Tiskaya M., Al‒Eesa NA., Wong FSL., Hill RG. Characterization of the bioactivity of two commercial composites. Dental Materials. 2019;35:1757‒68. [PubMed.]
- Featherstone JD. Prevention and reversal of dental caries: role of low level fluoride. Community Dent Oral Epidemiol. 1999;27:31‒40. [PubMed.]
- Rosin‒Grget K., Peros K., Sutej I., Basic K. The cariostatic mechanisms of fluoride. Acta Med Acad. 2013;42:179‒88. [PubMed.]
- Toumba KJ., Twetman S., Splieth C., Parnell C., Van Loveren C., et al. Guidelines on the use of fluoride for caries prevention in children: an updated EAPD policy document. European Archives of Paediatric Dentistry. 2019;20:507‒16. [PubMed.]
- Par M., Gubler A., Attin T., Tarle Z., Tarle A., et al. Ion release and hydroxyapatite precipitation of resin composites functionalized with two types of bioactive glass. Journal of Dentistry. 2022;118:103950. [PubMed.]
- Martínez‒Mier EA., Cury JA., Heilman JR., Katz BP., Levy SM., et al. Development of gold standard ion‒selective electrode‒based methods for fluoride analysis. Caries Res. 2011;45:3‒12. [PubMed.]
- Morales‒Valenzuela AA., Scougall‒Vilchis RJ., Lara‒Carrillo E., Garcia‒Contreras R., Hegazy‒Hassan W., et al. Enhancement of fluoride release in glass ionomer cements modified with titanium dioxide nanoparticles. Medicine (Baltimore). 2022;101:e31434. [PubMed.]
- Oliveira GL., Carvalho CN., Carvalho EM., Bauer J., Leal AMA. The Influence of Mixing Methods on the Compressive Strength and Fluoride Release of Conventional and Resin‒Modified Glass Ionomer Cements. Int J Dent. 2019;2019:6834931. [PubMed.]
- Patel S., Omid N., Zohoori FV., Maguire A., Waldron KJ., et al. Comparison of total ionic strength adjustment buffers III and IV in the measurement of fluoride concentration of teas. Nutrition and Health. 2018;24:111‒9. [PubMed.]
- Spano N., Bortolu S., Addis M., et al. An Analytical Protocol for the Differentiation and the Potentiometric Determination of Fluorine‒Containing Fractions in Bovine Milk. Molecules. 2023;28. [PubMed.]
- Venkateswarlu P. Evaluation of analytical methods for fluorine in biological and related materials. J Dent Res. 1990;69:514‒21. [PubMed.]
- Mullapudi VBK., Dheram K. A novel UV‒photolysis approach with acetone and isopropyl alcohol for the rapid determination of fluoride in organofluorine‒containing drugs by spectrophotometry. J Food Drug Anal. 2018;26:385‒92. [PubMed.]
- Prasad N., Pushpaangaeli B., Ram A., Maimanuku L. Fluoride concentration in drinking water samples in Fiji. Australian and New Zealand Journal of Public Health. 2018;42:372‒4. [PubMed.]
- Alebady MH., Hamama HH., Mahmoud SH. Effect of various surface coating methods on surface roughness., micromorphological analysis and fluoride release from contemporary glass ionomer restorations. BMC Oral Health. 2024;24. [PubMed.]
- Yardımcı B., Uzer A., Apak R. Spectrophotometric Fluoride Determination Using St. John's Wort Extract as a Green Chromogenic Complexant for Al(III). ACS Omega. 2022;7:45432‒42. [PubMed.]
- A GM., Diro A., Asere TG., Jado D., Melak F. Spectroscopic Determination of Fluoride Using Eriochrome Black T (EBT) as a Spectrophotometric Reagent from Groundwater. Int J Anal Chem. 2021;2021:2045491. [PubMed.]
- Manjunath V., John RP., Doddawad VG., Gehlot PM., Achar RR., et al. Measurement of Fluoride Ion Release From Restorative Material Using an Ion‒Selective Electrode and Ultraviolet‒Visible Light Spectrophotometer. J Int Soc Prev Community Dent. 2024;14:489‒96. [PubMed.]
- Cury JA., Caldarelli PG., Tenuta LM. Necessity to review the Brazilian regulation about fluoride toothpastes. Rev Saude Publica. 2015;49. [PubMed.]
- Sandulescu R., Florean E., Roman L., Mirel S., Oprean R., et al. Spectrophotometric determination of fluoride in dosage forms and dental preparations. J Pharm Biomed Anal. 1996;14:951‒8. [PubMed.]
- Adurty S., Karnam L., Chittor SN., Belliraj SK. Optical Sensing of Fluoride Through a Self‒Organized Fluorescent Ensemble of Quinizarin‒Al(III) Complex. J AOAC Int. 2016;99:1636‒41. [Ref.]
- Zolgharnein J., Shahrjerdi A., Azimi G., Ghasemi J. Spectrophotometric determination of trace amounts of fluoride using an Al‒xylenol orange complex as a colored reagent. Anal Sci. 2009;25:1249‒53. [PubMed.]
- Xu X., Jiang X., Shi M., Yin L. Mass spectrometry‒based techniques for single‒cell analysis. The Analyst. 2023;148:3690‒707. [PubMed.]
- Guo X., Wang X., Tian C., Dai J., Zhao Z., et al. Development of mass spectrometry imaging techniques and its latest applications. Talanta. 2023;264:124721. [PubMed.]
- Brands M., Gutbrod P., Dormann P. Lipid Analysis by Gas Chromatography and Gas Chromatography–Mass Spectrometry. Springer US. 2021:43‒57. [PubMed.]
- Du J., Sheng C., Wang Y., Zhang H., Jiang K. Determination of trace fluoride in water samples by silylation and gas chromatography/mass spectrometry analysis. Rapid Communications in Mass Spectrometry. 2021;35. [PubMed.]
- Biedermann M., Grob K. Advantages of comprehensive two‒dimensional gas chromatography for comprehensive analysis of potential migrants from food contact materials. Anal Chim Acta. 2019;1057:11‒7. [PubMed.]
- Lou C., Guo D., Wang N., Wu S., Zhang P., et al. Detection of trace fluoride in serum and urine by online membrane‒based distillation coupled with ion chromatography. J Chromatogr A. 2017;1500:145‒52. [PubMed.]
- Huang Y., Pang J., Zhang S., Huang W. Pretreatment methods in ion chromatography: A review. J Chromatogr A. 2024;1730:465162. [PubMed.]
- Reshetnyak VY., Nesterova OV., Admakin OI., Dobrokhotov DA., Avertseva IN., et al. Evaluation of free and total fluoride concentration in mouthwashes via measurement with ion‒selective electrode. BMC Oral Health. 2019;19. [PubMed.]
- Mondal NK. Diagnosis of Fluorosis by Analysis of Fluoride Content in Body Fluids Using Ion Selective Electrode Method. Springer International Publishing. 2021;121‒7. [PubMed.]
- Kissa E. Determination of inorganic fluoride in blood with a fluoride ion‒selective electrode. Clin Chem. 1987;33:253‒5. [PubMed.]
- Ignacio M., Curtzwiler GW., Early MR., Updegraff KM., Vorst KL. Ion Selective Electrode (ISE) Method for Determination of Total Fluorine and Total Organic Fluorine in Packaging Substrates. Methods Protoc. 2023;6. [Ref.]
- Radic J., Bralic M., Kolar M., Genorio B., Prkic A., et al. Development of the New Fluoride Ion‒Selective Electrode Modified with Fe(x)O(y) Nanoparticles. Molecules. 2020;25. [PubMed.]
- Wardak C., Morawska K., Pietrzak K. New Materials Used for the Development of Anion‒Selective Electrodes—A Review. Materials. 2023;16:5779. [PubMed.]
- Aguiar TR., Pinto CF., Cavalli V., Nobre‒dos‒Santos., Ambrosano GM., et al. Influence of the curing mode on fluoride ion release of self‒adhesive resin luting cements in water or during pH‒cycling regimen. Oper Dent. 2012;37:63‒70. [PubMed.]
- McCabe JF., Carrick TE., Sidhu SK. Determining low levels of fluoride released from resin based dental materials. Eur J Oral Sci. 2002;110:380‒4. [PubMed.]
- Wardak C., Morawska K., Pietrzak K. New Materials Used for the Development of Anion‒Selective Electrodes‒A Review. Materials (Basel). 2023;16. [PubMed.]
- Senthil Kumar R., Ravikumar N., Kavitha S., Mahalaxmi S., Jayasree R., et al. Nanochitosan modified glass ionomer cement with enhanced mechanical properties and fluoride release. Int J Biol Macromol. 2017;104:1860‒5. [PubMed.]
- Cheng L., Zhang K., Zhang N., Melo MAS., Weir MD., et al. Developing a New Generation of Antimicrobial and Bioactive Dental Resins. Journal of Dental Research. 2017;96:855‒63. [PubMed.]
Bioactive Properties Role on Material Development
How to Control Bacterial Progression
Factors Affecting Anticariogenicity
Evidence on the Bioactive Materials Effects on the Oral Biofilm and the Anticariogenic Effect
Methods of Evaluate Antibacterial Effect
Fluoride and Caries Prevention
Current Evidence on F Release of Tested Materials